JEE Main 2021 (18 March Shift 1)
For the following questions answer them individually
JEE Main 2021 (18 March Shift 1) - Question 51
________ grams of 3-Hydroxy propanal (MW = 74) must be dehydrated to produce 7.8 g of acrolein (MW = 56)(C$$_3$$H$$_4$$O) if the percentage yield is 64. (Round off to the Nearest Integer).
[Given: Atomic masses: C: 12.0u, H: 1.0u, O: 16.0u]
789
456
123
0.-
Clear All
JEE Main 2021 (18 March Shift 1) - Question 52
Complete combustion of 3 g of ethane gives x $$\times 10^{22}$$ molecules of water. The value of x is ________ (Round off to the Nearest Integer).
[Use: $$N_A = 6.023 \times 10^{23}$$; Atomic masses in u: C: 12.0; O: 16.0; H: 1.0]
789
456
123
0.-
Clear All
JEE Main 2021 (18 March Shift 1) - Question 53
AX is a covalent diatomic molecule where A and X are second row elements of periodic table. Based on Molecular orbital theory, the bond order of AX is 2.5. The total number of electrons in AX is ________. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
JEE Main 2021 (18 March Shift 1) - Question 54
For the reaction $$C_2H_6 \to C_2H_4 + H_2$$ the reaction enthalpy $$\Delta_r H$$ in kJ mol$$^{-1}$$ is ________. (Round off to the Nearest Integer).
[Given: Bond enthalpies in kJ mol$$^{-1}$$: C-C: 347, C=C: 611; C-H: 414, H-H: 436]
789
456
123
0.-
Clear All
JEE Main 2021 (18 March Shift 1) - Question 55
In order to prepare a buffer solution of pH 5.74, sodium acetate is added to acetic acid. If the concentration of acetic acid in the buffer is 1.0 M, the concentration of sodium acetate in the buffer is ________ M. (Round off to the Nearest Integer). [Given: pKa(acetic acid) = 4.74]
789
456
123
0.-
Clear All
JEE Main 2021 (18 March Shift 1) - Question 56
2 molal solution of a weak acid HA has a freezing point of 3.885°C. The degree of dissociation of this acid is ________ $$\times 10^{-3}$$. (Round off to the Nearest Integer). [Given: Molal depression constant of water = 1.85 K kg mol$$^{-1}$$, Freezing point of pure water = 0°C]
789
456
123
0.-
Clear All
JEE Main 2021 (18 March Shift 1) - Question 57
For the reaction $$2Fe^{3+}(aq) + 2I^-(aq) \to 2Fe^{2+}(aq) + I_2(s)$$, the magnitude of the standard molar free energy change, $$\Delta_r G_m^\circ$$ = -________ kJ (Round off to the Nearest Integer).
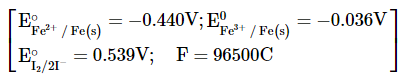
789
456
123
0.-
Clear All
JEE Main 2021 (18 March Shift 1) - Question 58
$$2NO(g) + Cl_2(g) \rightleftharpoons 2NOCl(s)$$
This reaction was studied at -10°C and the following data was obtained:
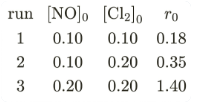
$$[NO]_{\circ}$$ and $$[Cl_{2}]_{\circ}$$ are the initial concentrations and $$r_{\circ}$$ is the initial reaction rate. The overall order of the reaction is ________. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
JEE Main 2021 (18 March Shift 1) - Question 59
The total number of unpaired electrons present in the complex K$$_3$$[Cr(oxalate)$$_3$$] is ________.
789
456
123
0.-
Clear All
JEE Main 2021 (18 March Shift 1) - Question 60
A reaction of 0.1 mole of Benzylamine with bromomethane gave 23 g of Benzyl trimethyl ammonium bromide. The number of moles of bromomethane consumed in this reaction are n $$\times 10^{-1}$$, when n = ________. (Round off to the Nearest Integer).
(Given: Atomic masses: C: 12.0 u, H: 1.0 u, N: 14.0 u, Br: 80.0 u)
789
456
123
0.-
Clear All


