NTA JEE Mains 9th April 2024 Shift 2
For the following questions answer them individually
NTA JEE Mains 9th April 2024 Shift 2 - Question 41
Match List I with List II:

Choose the correct answer from the options given below:
NTA JEE Mains 9th April 2024 Shift 2 - Question 42
Given below are two statements: Statement I : The higher oxidation states are more stable down the group among transition elements unlike p-block elements. Statement II : Copper can not liberate hydrogen from weak acids. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 9th April 2024 Shift 2 - Question 43
Match List I with List II:
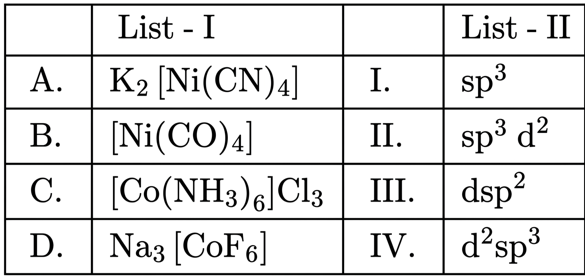
Choose the correct answer from the options given below:
NTA JEE Mains 9th April 2024 Shift 2 - Question 44
The coordination environment of $$Ca^{2+}$$ ion in its complex with $$EDTA^{4-}$$ is :
NTA JEE Mains 9th April 2024 Shift 2 - Question 45
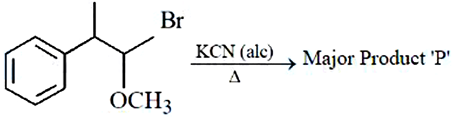
In the above reaction product '$$P$$' is :
NTA JEE Mains 9th April 2024 Shift 2 - Question 46
Match List I with List II:
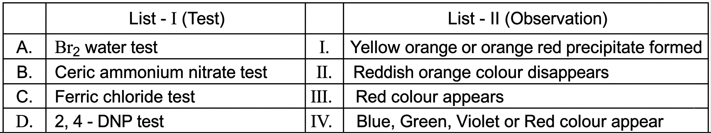
Choose the correct answer from the options given below:
NTA JEE Mains 9th April 2024 Shift 2 - Question 47
Which of the following compound can give positive iodoform test when treated with aqueous $$KOH$$ solution followed by potassium hypoiodite.
NTA JEE Mains 9th April 2024 Shift 2 - Question 48
Which of the following compounds will give silver mirror with ammoniacal silver nitrate? A. Formic acid B. Formaldehyde C. Benzaldehyde D. Acetone. Choose the correct answer from the options given below :
NTA JEE Mains 9th April 2024 Shift 2 - Question 49
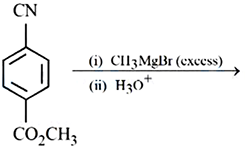
Major product of the following reaction is: A compound with $$CO_2CH_3$$ group reacts with (i) $$CH_3MgBr$$ (excess) (ii) $$H_3O^+$$
NTA JEE Mains 9th April 2024 Shift 2 - Question 50
The incorrect statement about Glucose is :


