NTA JEE Mains 9th April 2024 Shift 1
For the following questions answer them individually
NTA JEE Mains 9th April 2024 Shift 1 - Question 51
Molarity (M) of an aqueous solution containing $$x$$ g of anhyd. $$CuSO_4$$ in $$500$$ mL solution at $$32°C$$ is $$2 \times 10^{-1}$$ M. Its molality will be ______ $$\times 10^{-3}$$ m. (nearest integer). [Given density of the solution $$= 1.25$$ g/mL]
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 1 - Question 52
The total number of species from the following in which one unpaired electron is present, is _______
$$N_2, O_2, C_2^-, O_2^-, O_2^{2-}, H_2^+, CN^-, He_2^+$$
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 1 - Question 53
When equal volume of $$1$$ M HCl and $$1$$ M $$H_2SO_4$$ are separately neutralised by excess volume of $$1$$ M NaOH solution. $$x$$ and $$y$$ kJ of heat is liberated respectively. The value of $$y/x$$ is _______
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 1 - Question 54
The heat of solution of anhydrous $$CuSO_4$$ and $$CuSO_4 \cdot 5H_2O$$ are $$-70$$ kJ mol$$^{-1}$$ and $$+12$$ kJ mol$$^{-1}$$ respectively. The heat of hydration of $$CuSO_4$$ to $$CuSO_4 \cdot 5H_2O$$ is $$-x$$ kJ. The value of $$x$$ is ______ (nearest integer).
789
456
123
0.-
Clear All
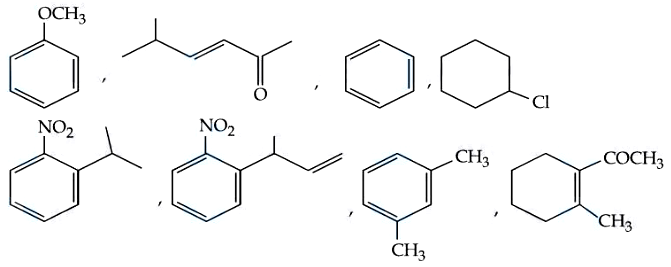
NTA JEE Mains 9th April 2024 Shift 1 - Question 55
How many compounds among the following compounds show inductive, mesomeric as well as hyperconjugation effects?
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 1 - Question 56
The standard reduction potentials at $$298$$ K for the following half cells are given below :
$$Cr_2O_7^{2-} + 14H^+ + 6e^- \rightarrow 2Cr^{3+} + 7H_2O$$, $$E° = 1.33$$ V
$$Fe^{3+}(aq) + 3e^- \rightarrow Fe$$, $$E° = -0.04$$ V
$$Ni^{2+}(aq) + 2e^- \rightarrow Ni$$, $$E° = -0.25$$ V
$$Ag^+(aq) + e^- \rightarrow Ag$$, $$E° = 0.80$$ V
$$Au^{3+}(aq) + 3e^- \rightarrow Au$$, $$E° = 1.40$$ V
Consider the given electrochemical reactions, The number of metal(s) which will be oxidized by $$Cr_2O_7^{2-}$$, in aqueous solution is ______
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 1 - Question 57
Given below are two statements : Statement I: The rate law for the reaction $$A + B \rightarrow C$$ is rate $$(r) = k[A]^2[B]$$. When the concentration of both A and B is doubled, the reaction rate is increased "$$x$$" times. Statement II :
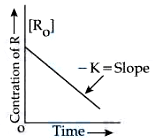
The figure is showing "the variation in concentration against time plot" for a "$$y$$" order reaction. The Value of $$x + y$$ is ______
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 1 - Question 58
Number of colourless lanthanoid ions among the following is ________
$$Eu^{3+}, Lu^{3+}, Nd^{3+}, La^{3+}, Sm^{3+}$$
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 1 - Question 59
Number of ambidentate ligands among the following is ______
$$NO_2^-, SCN^-, C_2O_4^{2-}, NH_3, CN^-, SO_4^{2-}, H_2O$$
789
456
123
0.-
Clear All
NTA JEE Mains 9th April 2024 Shift 1 - Question 60
Total number of essential amino acid among the given list of amino acids is _______ Arginine, Phenylalanine, Aspartic acid, Cysteine, Histidine, Valine, Proline
789
456
123
0.-
Clear All


