NTA JEE Mains 9th April 2024 Shift 1
For the following questions answer them individually
NTA JEE Mains 9th April 2024 Shift 1 - Question 31
Compare the energies of following sets of quantum numbers for multielectron system. (A) $$n = 4, l = 1$$ (B) $$n = 4, l = 2$$ (C) $$n = 3, l = 1$$ (D) $$n = 3, l = 2$$ (E) $$n = 4, l = 0$$. Choose the correct answer from the options given below :
NTA JEE Mains 9th April 2024 Shift 1 - Question 32
Given below are two statements : Statement (I) : The oxidation state of an element in a particular compound is the charge acquired by its atom on the basis of electron gain enthalpy consideration from other atoms in the molecule. Statement (II) : $$p\pi - p\pi$$ bond formation is more prevalent in second period elements over other periods. In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Mains 9th April 2024 Shift 1 - Question 33
In which one of the following pairs the central atoms exhibit $$sp^2$$ hybridization ?
NTA JEE Mains 9th April 2024 Shift 1 - Question 34
Identify the incorrect statements regarding primary standard of titrimetric analysis. (A) It should be purely available in dry form. (B) It should not undergo chemical change in air. (C) It should be hygroscopic and should react with another chemical instantaneously and stoichiometrically. (D) It should be readily soluble in water. (E) $$KMnO_4$$ & $$NaOH$$ can be used as primary standard. Choose the correct answer from the options given below :
NTA JEE Mains 9th April 2024 Shift 1 - Question 35
Methods used for purification of organic compounds are based on :
NTA JEE Mains 9th April 2024 Shift 1 - Question 36
Correct order of basic strength of Pyrrole,
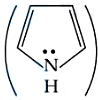
Pyridine,
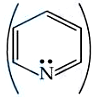
and Piperidine
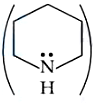
is:
NTA JEE Mains 9th April 2024 Shift 1 - Question 37
For the given compounds, the correct order of increasing $$pK_a$$ value :
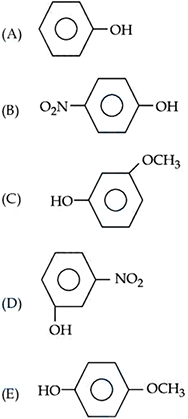
Choose the correct answer from the options given below :
NTA JEE Mains 9th April 2024 Shift 1 - Question 38
Relative stability of the contributing structures is
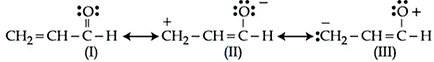
NTA JEE Mains 9th April 2024 Shift 1 - Question 39
Identify the product A and product B in the following set of reactions.
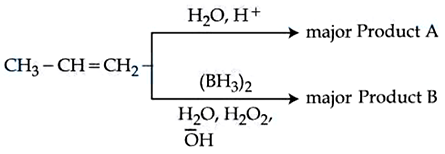
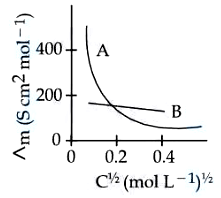
NTA JEE Mains 9th April 2024 Shift 1 - Question 40
The molar conductivity for electrolytes A and B are plotted against $$C^{1/2}$$ as shown below. Electrolytes A and B respectively are