NTA JEE Mains 8th April Shift 2 2026
For the following questions answer them individually
NTA JEE Mains 8th April Shift 2 2026 - Question 61
Given below are two statements for catalytic properties of transition metals.
Statement I : First row transition metals which act as catalyst utilise their 3d electrons only for formation of bonds between reactant molecules and atoms on the surface of catalyst.
Statement II : There is increase in the concentration of reactants on the surface of catalyst which strengthens the bonds in reacting molecules.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 8th April Shift 2 2026 - Question 62
Given below are two statements :
Statement I : Vapours of the liquid with higher boiling point condense before vapours of the liquid with lower boiling points in fractional distillation.
Statement II : The vapours rising up in the fractionating column become richer in high boiling component of the mixture.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 8th April Shift 2 2026 - Question 63
The major product of which of the following reaction is not obtained by rearrangement reaction?
NTA JEE Mains 8th April Shift 2 2026 - Question 64
The total number of aromatic compounds/species from the following is
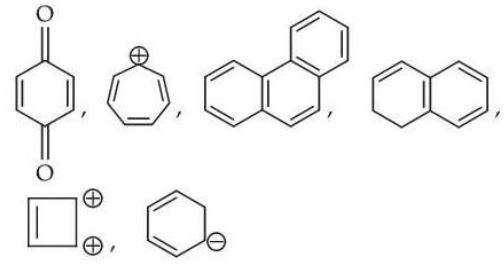
NTA JEE Mains 8th April Shift 2 2026 - Question 65
n-Butane on monochlorination under photochemical condition gives an optically active compound "P". "P" on further chlorination gives dichloro compounds. The number of dichloro compounds obtained (ignore stereoisomers) is :
NTA JEE Mains 8th April Shift 2 2026 - Question 66
Given below are two statements :
Statement I : Due to increase in van der Waals forces, the order of boiling points is CH$$_3$$CH$$_2$$CH$$_2$$I > CH$$_3$$CH$$_2$$I > CH$$_3$$I.
Statement II : As
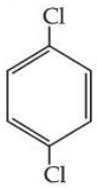
is more symmetric, its melting point is higher than
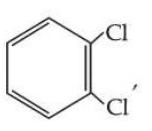
, however its boiling point is lower than
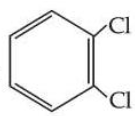
.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 8th April Shift 2 2026 - Question 67
Consider the following reaction.
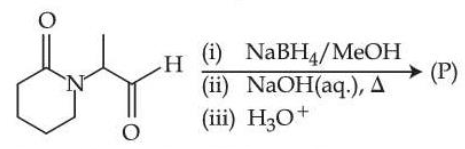
The major product (P) formed is :
NTA JEE Mains 8th April Shift 2 2026 - Question 68
Which statements are True?
A. In Hoffmann bromamide degradation, 4 moles of NaOH and 2 moles of Br$$_2$$ are consumed per mole of an amide
B. Hoffmann bromamide reaction is not given by alkyl amides.
C. Primary amines can be synthesized by Hoffmann bromamide degradation.
D. Secondary amide on reaction with Br$$_2$$ and NaOH will give secondary amine.
E. The by-products of Hoffmann degradation are Na$$_2$$CO$$_3$$, NaBr and H$$_2$$O.
Choose the correct answer from the options given below :
NTA JEE Mains 8th April Shift 2 2026 - Question 69
The incorrect statement from the following with respect to carbohydrates is :
NTA JEE Mains 8th April Shift 2 2026 - Question 70
Which of the following amino acid will give violet coloured complex with neutral ferric chloride solution?


