NTA JEE Mains 8th April Shift 2 2026 - Chemistry
For the following questions answer them individually
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 51
Match List - I with List - II.

Choose the correct answer from the options given below :
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 52
Given below are two statements :
Given : Molar mass of C, H, O, Cl are 12, 1, 16 and 35.5 g mol$$^{-1}$$, respectively
Statement I : In 30% (w/w) solution of methanol in CCl$$_4$$(at T K), the mole fraction of CCl$$_4$$ is equal to 0.33.
Statement II : Mixture of methanol and CCl$$_4$$ shows positive deviation from Raoult's law.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 53
Bromine trifluoride autoionizes to form $$\text{BrF}_2^+$$ and $$\text{BrF}_4^-$$. The shapes of the cation and anion are respectively __________, and __________.
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 54
Which of the following statements are not correct?
A. For water, magnitude of $$K_b$$ is more than the magnitude of $$K_f$$.
B. The elevation in boiling point of water when a non-volatile solute is added to it is larger in magnitude than its depression in freezing point.
C. Osmotic pressure measurement is preferred over any other colligative property to determine molar mass of proteins and polymers.
D. The dimerised form of benzoic acid in benzene is
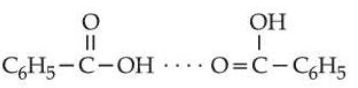
Choose the correct answer from the options given below :
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 55
Consider the following reactions in which all the reactants and products are present in gaseous state
$$2xy \rightleftharpoons x_2 + y_2 \quad K_1 = 2.5 \times 10^5$$
$$xy + \frac{1}{2}z_2 \rightleftharpoons xyz \quad K_2 = 5 \times 10^{-3}$$
The value of $$K_3$$ for the equilibrium $$\frac{1}{2}x_2 + \frac{1}{2}y_2 + \frac{1}{2}z_2 \rightleftharpoons xyz$$ is :
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 56
Given at 298 K :
$$E^\ominus_{\text{Fe}^{2+}/\text{Fe}} = X$$ Volt
$$E^\ominus_{\text{Fe}^{3+}/\text{Fe}} = Y$$ Volt
The $$E^\ominus_{\text{Fe}^{3+}/\text{Fe}^{2+}}$$ in Volt at 298 K is given by :
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 57
Given below are two statements :
$$R = 8.314$$ J K$$^{-1}$$ mol$$^{-1}$$ and 1 cal = 4.2 J
Statement I : When $$E_a = 12.6$$ kcal/mol, the room temperature rate constant is doubled by a 10 $$^\circ$$C increase in temperature (298 K to 308 K)
Statement II : For a first order reactions $$A \to B$$,
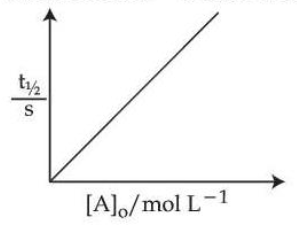
Here $$[A]_0$$ is the initial concentration of A and $$t_{1/2}$$ is half life of reaction.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 58
Match List - I with List - II.
List - I (Electronic configuration of neutral atom where n = 2) List - II (1st Ionization Energy in kJ mol$$^{-1}$$)
A. ns$$^2$$ I. 2080
B. ns$$^2$$np$$^1$$ II. 899
C. ns$$^2$$np$$^3$$ III. 800
D. ns$$^2$$np$$^6$$ IV. 1402
Choose the correct answer from the options given below :
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 59
Find the correct statements related to group 15 hydrides.
A. Reducing nature increases from NH$$_3$$ to BiH$$_3$$
B. Tendency to donate lone pair of electrons decreases from NH$$_3$$ to BiH$$_3$$
C. The stability of hydrides decreases from NH$$_3$$ to BiH$$_3$$
D. HEH bond angle decreases from NH$$_3$$ to SbH$$_3$$ (E = Elements of group 15)
Choose the correct answer from the options given below :
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 60
Given below are two statements :
Statement I : The number of pairs among [Ti$$^{4+}$$, V$$^{2+}$$], [V$$^{2+}$$, Mn$$^{2+}$$], [Mn$$^{2+}$$, Fe$$^{3+}$$] and [V$$^{2+}$$, Cr$$^{2+}$$] in which both ions are coloured is 3.
Statement II : The number of pairs among [La$$^{3+}$$, Yb$$^{2+}$$], [Lu$$^{3+}$$, Ce$$^{4+}$$] and [Ac$$^{3+}$$, Lr$$^{3+}$$] ions in which both are diamagnetic is 3.
In the light of the above statements, choose the correct from the options given below :
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 61
Given below are two statements for catalytic properties of transition metals.
Statement I : First row transition metals which act as catalyst utilise their 3d electrons only for formation of bonds between reactant molecules and atoms on the surface of catalyst.
Statement II : There is increase in the concentration of reactants on the surface of catalyst which strengthens the bonds in reacting molecules.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 62
Given below are two statements :
Statement I : Vapours of the liquid with higher boiling point condense before vapours of the liquid with lower boiling points in fractional distillation.
Statement II : The vapours rising up in the fractionating column become richer in high boiling component of the mixture.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 63
The major product of which of the following reaction is not obtained by rearrangement reaction?
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 64
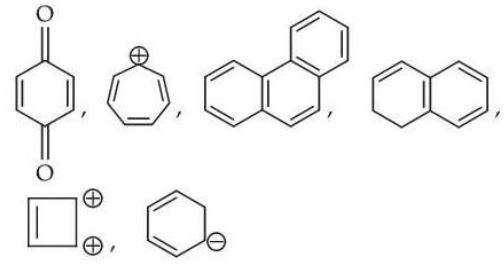
The total number of aromatic compounds/species from the following is
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 65
n-Butane on monochlorination under photochemical condition gives an optically active compound "P". "P" on further chlorination gives dichloro compounds. The number of dichloro compounds obtained (ignore stereoisomers) is :
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 66
Given below are two statements :
Statement I : Due to increase in van der Waals forces, the order of boiling points is CH$$_3$$CH$$_2$$CH$$_2$$I > CH$$_3$$CH$$_2$$I > CH$$_3$$I.
Statement II : As
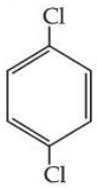
is more symmetric, its melting point is higher than
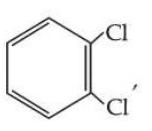
, however its boiling point is lower than
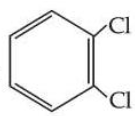
.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 67
Consider the following reaction.
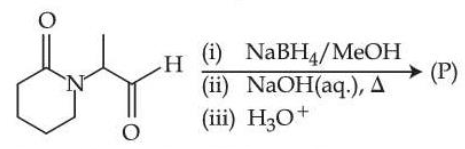
The major product (P) formed is :
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 68
Which statements are True?
A. In Hoffmann bromamide degradation, 4 moles of NaOH and 2 moles of Br$$_2$$ are consumed per mole of an amide
B. Hoffmann bromamide reaction is not given by alkyl amides.
C. Primary amines can be synthesized by Hoffmann bromamide degradation.
D. Secondary amide on reaction with Br$$_2$$ and NaOH will give secondary amine.
E. The by-products of Hoffmann degradation are Na$$_2$$CO$$_3$$, NaBr and H$$_2$$O.
Choose the correct answer from the options given below :
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 69
The incorrect statement from the following with respect to carbohydrates is :
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 70
Which of the following amino acid will give violet coloured complex with neutral ferric chloride solution?
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 71
Number of paramagnetic complexes among the following is __________.
$$[\text{MnBr}_4]^{2-}$$, $$[\text{NiCl}_4]^{2-}$$, $$[\text{Ni(CN)}_4]^{2-}$$, $$[\text{Ni(CO)}_4]$$, $$[\text{CoF}_6]^{3-}$$, $$[\text{Fe(CN)}_6]^{4-}$$, $$[\text{Mn(CN)}_6]^{3-}$$, $$[\text{Ti(CN)}_6]^{3-}$$, $$[\text{Cu(H}_2\text{O)}_6]^{2+}$$, $$[\text{Co(C}_2\text{O}_4)_3]^{3-}$$
789
456
123
0.-
Clear All
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 72
'$$x$$' is the product which is obtained from benzene by reacting it with carbon monoxide and hydrogen chloride in the presence of cuprous chloride. '$$y$$' is the major product obtained from the benzene by reacting it with ethanoyl chloride in the presence of anhydrous AlCl$$_3$$. Product (major) obtained by heating $$x$$ and $$y$$ in the presence of alkali is $$z$$. Total number of $$\pi$$ (pi) electrons in $$z$$ is __________.
789
456
123
0.-
Clear All
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 73
Consider two radiations of wavelengths
1. $$\lambda_1 = 2000$$ $$\text{\AA}$$
2. $$\lambda_2 = 6000$$ $$\text{\AA}$$
The ratio of the energies of these two radiations $$\left(\frac{E_1}{E_2}\right)$$ is __________ (Nearest integer).
789
456
123
0.-
Clear All
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 74
Consider the reaction
$$2\text{H}_2\text{S(g)} + 3\text{O}_2\text{(g)} \to 2\text{H}_2\text{O(l)} + 2\text{SO}_2\text{(g)}$$
The magnitude of enthalpy change for the reaction in kJ mol$$^{-1}$$ is __________. (Nearest integer)
Given :
$$\Delta_f H^\ominus(\text{H}_2\text{S}) = -20.1$$ kJ mol$$^{-1}$$
$$\Delta_f H^\ominus(\text{H}_2\text{O}) = -286.0$$ kJ mol$$^{-1}$$
$$\Delta_f H^\ominus(\text{SO}_2) = -297.0$$ kJ mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Mains 8th April Shift 2 2026 - Chemistry - Question 75
Solid carbon, CaO and CaCO$$_3$$ are mixed and allowed to attain equilibrium at T K.
$$\text{CaCO}_3\text{(s)} \rightleftharpoons \text{CaO(s)} + \text{CO}_2\text{(g)} \quad K_{p_1} = 0.08$$ atm
$$\text{C(s)} + \text{CO}_2\text{(g)} \rightleftharpoons 2\text{CO(g)} \quad K_{p_2} = 2$$ atm
The partial pressure of CO is __________ $$\times 10^{-1}$$ atm
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




