NTA JEE Mains 6th April Shift 2 2026 - Chemistry
For the following questions answer them individually
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 51
Which of the following contain the same number of atoms?
(Given : Molar mass in g mol$$^{-1}$$ of H, He, O and S are 1, 4, 16 and 32 respectively)
A. 2 g of O$$_2$$ gas
B. 4 g of SO$$_2$$ gas
C. 1400 mL of O$$_2$$ at STP
D. 0.05 L of He at STP
E. 0.0625 mol of H$$_2$$ gas
Choose the correct answer from the options given below :
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 52
The Bohr radius of a hydrogen like species is 70.53 pm. The species and the stationary state (n) are respectively (Given : Hydrogen atom Bohr radius is 52.9 pm)
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 53
Given below are two statements :
Statement I : The number of compounds among SO$$_2$$, SO$$_3$$, SF$$_4$$, SF$$_6$$ and H$$_2$$S in which sulphur does not obey the Octet rule is 3.
Statement II : Among [H$$_2$$O, ClF$$_3$$, SF$$_4$$], [NH$$_3$$, BrF$$_5$$, SF$$_4$$], [BrF$$_5$$, ClF$$_3$$, XeF$$_4$$] and [XeF$$_4$$, ClF$$_3$$, H$$_2$$O], the number of sets in which all the molecules have one lone pair of electrons on the central atom is 1.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 54
Match List - I with List - II.
Given $$V_1$$ and $$V_2$$ are initial and final volumes respectively.

Choose the correct answer from the options given below :
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 55
Given below are two statements :
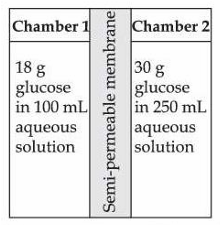
Statement I : H$$_2$$O molecules move from the chamber 1 to chamber 2.
Statement II : The osmotic pressure of a solution prepared by dissolving 50 mg of potassium sulphate (molar mass = 174 g/mol) in 2 L of water (at 27 °C) is 0.0107 bar. (Given: R = 0.083 dm$$^3$$ bar K$$^{-1}$$ mol$$^{-1}$$ and assume complete dissociation of electrolyte)
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 56
Given is a concentrated solution of a weak electrolyte $$A_xB_y$$ of concentration 'c' and dissociation constant 'K'. The degree of dissociation is given by :
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 57
For a general redox reaction
Anode: $$\text{Red}_1 \to \text{Ox}_1^{n_1+} + n_1 e^-$$
Cathode: $$\text{Ox}_2 + n_2 e^- \to \text{Red}_2^{n_2-}$$
Which of the following statement is **incorrect** ?
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 58
In a period, the first ionisation enthalpy of the element at extreme left and the negative electron gain enthalpy of the extreme right element, except noble gases, are respectively.
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 59
Given below are two statements :
Statement I : $$F_2O < H_2O < Cl_2O$$ is the correct trend in terms of bond angle.
Statement II : SiF$$_4$$, SnF$$_4$$ and PbF$$_4$$ are ionic in nature.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 60
The correct order of first ($$\Delta_i H_1$$) and second ($$\Delta_i H_2$$) ionisation enthalpy values of Cr and Mn are :
A. $$\Delta_i H_1$$ : Cr > Mn
B. $$\Delta_i H_2$$ : Cr > Mn
C. $$\Delta_i H_1$$ : Mn > Cr
D. $$\Delta_i H_2$$ : Mn > Cr
Choose the correct answer from the options given below :
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 61
Which of the following sequences of hybridisation, geometry and magnetic nature are correct for the given coordination compounds?
A. $$[NiCl_4]^{2-}$$ -- sp$$^3$$, tetrahedral, paramagnetic
B. $$[Ni(NH_3)_6]^{2+}$$ -- sp$$^3$$d$$^2$$, octahedral, paramagnetic
C. $$[Ni(CO)_4]$$ -- sp$$^3$$, tetrahedral, paramagnetic
D. $$[Ni(CN)_4]^{2-}$$ -- dsp$$^2$$, square planar, diamagnetic
Choose the correct answer from the options given below :
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 62
Given below are two statements :
Statement I : A mixture of C$$_{12}$$H$$_{22}$$O$$_{11}$$ (sugar) and NaCl can be separated by dissolving sugar in alcohol, due to differential solubility.
Statement II : Rose essence from rose petals is separated by steam distillation due to its high volatility and insolubility in H$$_2$$O.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 63
Shown below is the structure of methyl acetate with three different $$\alpha$$, $$\beta$$ and $$\gamma$$ carbon-oxygen bonds.
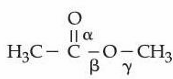
The correct order of bond lengths of these bonds is :
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 64
'x' is the product which is obtained by the hydrolysis of prop-1-yne in the presence of mercuric sulphate under dilute acidic medium at 333 K. 'y' is the product which is obtained by the reaction of ethane nitrile with methyl magnesium bromide in dry ether followed by hydrolysis. IUPAC name of product obtained from 'x' and 'y' in the presence of barium hydroxide followed by heating is :
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 65
An optically active alkyl bromide C$$_4$$H$$_9$$Br, reacts with ethanolic KOH to form major compound [A] which reacts with bromine to give compound [B]. Compound [B] reacts with ethanolic KOH and sodamide to give compound [C]. One molecule of water adds to compound [C] on warming with mercuric sulphate and dilute sulphuric acid at 333 K to form compound [D]. The functional group in compound D will be confirmed by :
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 66
Consider the following reaction.
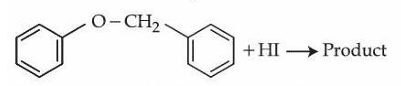
Statement I : In the above reaction, product formed will be a mixture of benzyl alcohol and iodobenzene.
Statement II : In the above reaction, the $$-O-CH_2-$$ bond is cleaved to give the product.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 67
Consider the following organic reaction sequence. Choose the final product (X) from the following (consider the major product in all intermediate reactions)
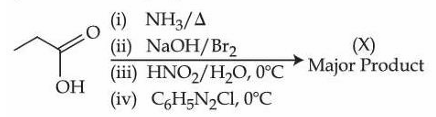
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 68
The number of compounds from the following which can undergo reaction with Br$$_2$$/KOH (alcoholic) to give respective products and these respective products can also be obtained separately by Gabriel phthalimide reaction is :
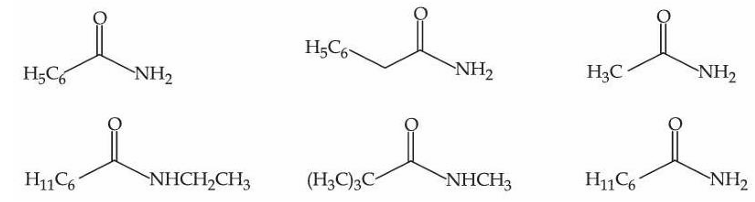
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 69
Consider the following reactions. Total number of electrons in the $$\pi$$ bonds and lone pair of electrons in the product (X) is :

NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 70
Treatment of a gas 'X' with a freshly prepared ferrous sulphate solution gives a compound 'Y' as a brown ring. The compounds X and Y are.
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 71
An excess of AgNO$$_3$$ is added to 100 mL of a 0.05 M solution of tetraaquadichloridochromium (III) chloride. The number of moles of AgCl precipitated will be __________ $$\times 10^{-3}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 72
An alkane (Y) requires 8 moles of oxygen for complete combustion and on chlorination with Cl$$_2$$/h$$\nu$$, (Y) gives only one monochlorinated product (Z). The total number of primary carbon atoms in (Y) is __________.
789
456
123
0.-
Clear All
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 73
500 mL of 0.2 M MnO$$_4^-$$ solution in basic medium when mixed with 500 mL of 1.5 M KI solution, oxidises iodide ions to liberate molecular iodine. This liberated iodine is then titrated with a standard $$x$$ M thiosulphate solution in presence of starch till the end point. If 300 mL of thiosulphate was consumed, then the value of $$x$$ is __________.
789
456
123
0.-
Clear All
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 74
In a closed flask at 600 K, one mole of $$X_2Y_4(g)$$ attains equilibrium as given below : $$$X_2Y_4(g) \rightleftharpoons 2XY_2(g)$$$ At equilibrium, 75% $$X_2Y_4(g)$$ was dissociated and the total pressure is 1 atm. The magnitude of $$\Delta_r G^{\ominus}$$ (in kJ mol$$^{-1}$$) at this temperature is __________. (Nearest Integer) (Given : R = 8.3 J mol$$^{-1}$$ K$$^{-1}$$; ln 10 = 2.3, log 2 = 0.3, log 3 = 0.48, log 5 = 0.69, log 7 = 0.84)
789
456
123
0.-
Clear All
NTA JEE Mains 6th April Shift 2 2026 - Chemistry - Question 75
Decomposition of a hydrocarbon follows the equation $$k = (5.5 \times 10^{11} s^{-1}) e^{\frac{-28000K}{T}}$$. The activation energy of reaction is __________ kJ mol$$^{-1}$$. (Nearest Integer) Given : R = 8.3 J K$$^{-1}$$ mol$$^{-1}$$
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




