NTA JEE Mains 6th April Shift 1 2026 - Chemistry
For the following questions answer them individually
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 51
An oxide of iron contains 69.9% iron, its empirical formula, is:
(Given: Molar mass of Fe and O are 56 and 16 g mol$$^{-1}$$ respectively.)
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 52
If shortest wavelength of hydrogen atom in Lyman series is $$x$$, then longest wavelength in Balmer series of He$$^-$$ is:
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 53
Match the LIST-I with LIST-II
| List-I Orbital | List-II Radial nodes and nodal plane | ||
|---|---|---|---|
| A. | 2s | I. | 1 Radial node + two nodal planes |
| B. | 3s | II. | 1 Radial node + one nodal plane |
| C. | 3p | III. | 2 Radial nodes + No nodal plane |
| D. | 4d | IV. | 1 Radial node + No nodal plane |
Choose the correct answer from the options given below:
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 54
The pairs among
$$A = [SO_3^{2-}, CO_3^{2-}]$$, $$B = [O_2^{2-}, F_2]$$, $$C = [CN^-, CO]$$, $$D = [NH_3, H_3O^+]$$ and $$E = [MnO_4^{2-}, CrO_4^{2-}]$$ that do not have similar Lewis dot structure are:
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 55
Arrange the following isothermal processes in order of the magnitude of the work (p - V) involved between states 1 and 2.
A. Expansion in single stage $$w_A$$
B. Expansion in multi stages $$w_B$$
C. Compression in single stage $$w_C$$
D. Compression in multi stages $$w_D$$
Choose the correct option.
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 56
When 0.25 moles of a non-volatile, non-ionizable solute was dissolved in 1 mole of a solvent the vapor pressure of solution was $$x$$ % of vapor pressure of pure solvent. What is $$x$$ %?
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 57
One mole each of He and A(g) are taken in a 10 L closed flask and heated to 400 K to establish the following equilibrium.
A(g) $$\rightleftharpoons$$ B(g)
K$$_c$$ for this reaction at 400 K is 4.0. The partial pressures (in atm) of He and B(g) are respectively (at equilibrium)
(Assume He, A(g) and B(g) behave as ideal gases)
(Given: R = 0.082 L atm K$$^{-1}$$ mol$$^{-1}$$)
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 58
Consider the following data.
| Electrolyte | $$\Lambda^\circ_m$$ (S cm$$^2$$ mol$$^{-1}$$) |
|---|---|
| BaCl$$_2$$ | $$x_1$$ |
| H$$_2$$SO$$_4$$ | $$x_2$$ |
| HCl | $$x_3$$ |
BaSO$$_4$$ is sparingly soluble in water. If the conductivity of the saturated BaSO$$_4$$ solution is $$x$$ S cm$$^{-1}$$ then the solubility product of BaSO$$_4$$ can be given as
(Here $$\Lambda_m = \Lambda^\circ_m$$)
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 59
Given below are two statements:
Statement I: Aluminium is more electropositive than thallium as the standard electrode potential value of E$$^\circ_{Al^{3+}/Al}$$ is negative and E$$^\circ_{Tl^{3+}/Tl}$$ is positive.
Statement II: The sum of first three ionization enthalpies of boron is very high when compared to that of aluminium. Due to this reason boron forms covalent compounds only and aluminium forms Al$$^{3+}$$ ion.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 60
The correct statements among the following are.
A. Basic vanadium oxide is used in the manufacture of H$$_2$$SO$$_4$$.
B. The spin-only magnetic moment value of the transition metal halide employed in Ziegler-Natta polymerization is 2.84 BM.
C. The p-block metal compound employed in Ziegler-Natta polymerization has the metal in +3 oxidation state.
D. The number of electrons present in the outer most 'd' orbital of metal halide employed in Wacker process is 8.
Choose the correct answer from the options given below:
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 61
Match the LIST-I with LIST-II
| List-I Electronic configuration of tetrahedral metal ion | List-II Crystal Field Stabilization Energy ($$\Delta_t$$) | ||
|---|---|---|---|
| A. | d$$^2$$ | I. | -0.6 |
| B. | d$$^4$$ | II. | -0.8 |
| C. | d$$^6$$ | III. | -1.2 |
| D. | d$$^8$$ | IV. | -0.4 |
Choose the correct answer from the options given below:
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 62
Which of the following are true about the energy of the given d-orbitals of a tetrahedral complex?
A. $$d_{xy} = d_{xz} > d_{x^2-y^2}$$
B. $$d_{xy} = d_{yz} > d_{z^2}$$
C. $$d_{x^2-y^2} > d_{z^2} > d_{xz}$$
D. $$d_{x^2-y^2} = d_{z^2} < d_{xz}$$
Choose the correct answer from the given below:
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 63
R$$_f$$ value for 2-methylpropene in a solvent system (Ethyl acetate + ether) is 0.42. 2-methylpropene is treated with dilute H$$_2$$SO$$_4$$ to give major organic product (X). R$$_f$$ value for (X) in the same solvent system under identical condition will be:
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 64
Given below are two statements:
Statement I: 2,6-diethylcyclohexanone and 6-methyl-2-n-propylcyclohexanone are metamers.
Statement II: 2,2,6,6-tetramethylcyclohexanone exhibits keto-enol tautomerism.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 65
Given below are two statements:
Statement I: Methane can be prepared by decarboxylation of sodium ethanoate, Kolbe's electrolysis of sodium acetate and reaction of CH$$_3$$MgBr with water.
Statement II: Methane cannot be prepared from unsaturated hydrocarbons and by Wurtz reaction.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 66
Given below are two statements:
Statement I: 3-phenylpropene reacts with HBr and gives secondary alkyl bromide having a chiral carbon atom as the major product.
Statement II: Aryl chlorides and aryl cyanides can be prepared by Sandmeyer reaction as well as Gattermann reaction.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 67
Consider the following sequence of reactions
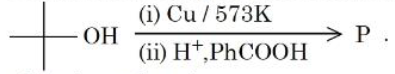
The major product P is:
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 68
Arrange the following compounds according to increasing order of boiling points.
n-C$$_4$$H$$_9$$OH (A), n-C$$_4$$H$$_9$$NH$$_2$$ (B), n-C$$_4$$H$$_{10}$$ (C) and C$$_2$$H$$_5$$NHC$$_2$$H$$_5$$ (D).
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 69
Match the LIST-I with LIST-II
| List-I Deficiency Disease | List-II Vitamin | ||
|---|---|---|---|
| A. | Scurvy | I. | Pyridoxine |
| B. | Convulsions | II. | Vitamin A |
| C. | Cheilosis | III. | Ascorbic Acid |
| D. | Xerophthalmia | IV. | Riboflavin |
Choose the correct answer from the options given below:
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 70
Match the LIST-I with LIST-II
| List-I Amino acid | List-II Positive reaction/Test for functional group present in side chain of amino acid | ||
|---|---|---|---|
| A. | Glutamine | I. | Hinsberg's test |
| B. | Lysine | II. | Neutral FeCl$$_3$$ test |
| C. | Tyrosine | III. | Ceric ammonium nitrate test |
| D. | Serine | IV. | Hoffman bromamide degradation |
Choose the correct answer from the options given below:
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 71
First and second ionization enthalpies of lithium are 520 kJ mol$$^{-1}$$ and 7297 kJ mol$$^{-1}$$ respectively. Energy required to convert 3.5 mg lithium (g) into Li$$^{2+}$$(g) [Li(g) $$\to$$ Li$$^{2+}$$(g)] is _______ kJ mol$$^{-1}$$. (nearest integer)
[Molar mass of Li = 7 g mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 72
Consider the following sequence of reactions.
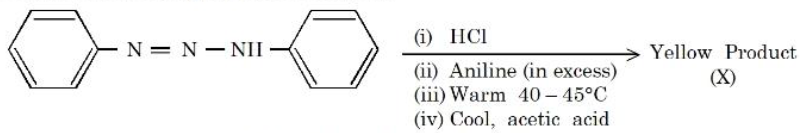
The percentage of nitrogen in the yellow product (X) formed is _______ %. (Nearest Integer)
(Given Molar mass in g mol$$^{-1}$$ H:1, C:12, N:14)
789
456
123
0.-
Clear All
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 73
4.7 g of phenol is heated with Zn to give product X. If this reaction goes to 60% completion then the number of moles of compound X formed will be _______ $$\times 10^{-2}$$. (Nearest Integer)
(Given molar mass in g mol$$^{-1}$$: H:1, C:12, O:16)
789
456
123
0.-
Clear All
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 74
Sucrose hydrolyses in acidic medium into glucose and fructose by first order rate law with $$t_{1/2} = 3$$ hour. The percentage of sucrose remaining after 6 hours is _______. (Nearest integer)
(Given: log 2 = 0.3010 and log 3 = 0.4771)
789
456
123
0.-
Clear All
NTA JEE Mains 6th April Shift 1 2026 - Chemistry - Question 75
Consider the reaction X $$\rightleftharpoons$$ Y at 300 K. If $$\Delta H^\theta$$ and K are 28.40 kJ mol$$^{-1}$$ and $$1.8 \times 10^{-7}$$ at the same temperature, then the magnitude of $$\Delta S^\theta$$ for the reaction in J K$$^{-1}$$ mol$$^{-1}$$ is _______. (Nearest integer)
(Given: R = 8.3 J K$$^{-1}$$ mol$$^{-1}$$, ln 10 = 2.3, log 3 = 0.48, log 2 = 0.30)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




