NTA JEE Mains 5th April Shift 2 2026 - Chemistry
For the following questions answer them individually
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 51
What volume of hydrogen gas at STP would be liberated by action of 50 mL of $$H_2SO_4$$ of 50% purity (density = 1.3 g mL$$^{-1}$$) on 20 g of zinc?
Given : Molar mass of H, O, S, Zn are 1, 16, 32, 65 g mol$$^{-1}$$ respectively.
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 52
Which of the following statement(s) is/are true?
$$\textbf{A.}$$ If two orbitals have the same value of $$(n + l)$$, the orbital with lower value of $$n$$ will have lower energy.
$$\textbf{B.}$$ Energies of the orbitals in the same subshell increase with increase in atomic number.
$$\textbf{C.}$$ The size of $$2p_x$$ orbital is less than the size of $$3p_x$$ orbital.
$$\textbf{D.}$$ Among $$5f$$, $$6s$$, $$4d$$, $$5p$$ and $$5d$$ orbitals, none of the orbitals have 2 radial nodes.
Choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 53
The covalent radii of atoms A and B are $$r_A$$ and $$r_B$$ respectively. The covalent bond length and total length of $$AB$$ molecule are respectively :
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 54
Consider the following data for the reaction
$$\text{X}_2(g) + \text{Y}_2(g) \rightleftharpoons 2\text{XY}(g)$$
at 600 K. The $$\Delta_r G^\circ$$ (in $$kJ/mol^{-1}$$) for the reaction is :

NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 55
The correct order of molar heat capacities measured at 298 K and 1 bar is :
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 56
The reaction $$A(g) \rightleftharpoons B(g) + C(g)$$ was initiated with the amount `$$a$$` of $$A(g)$$. At equilibrium it is found that the amount of $$A(g)$$ remaining is $$(a - x)$$ at a total pressure of $$p$$.
The equilibrium constant $$K_p$$ of the reaction can be calculated from the expression:
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 57
One half cell in a voltaic cell is constructed by dipping silver rod in $$\text{AgNO}_3$$ solution of unknown concentration, other half cell is Zn rod dipped in 1 molar solution of $$\text{ZnSO}_4$$. A voltage of $$1.60\,\text{V}$$ is measured at $$298\,\text{K}$$ for this cell. What is the concentration of $$\text{Ag}^+$$ ions used in terms of $$\log x$$ $$(x = [\text{Ag}^+])$$?
$$E^\ominus_{\text{Zn}^{2+}/\text{Zn}} = -0.76\,\text{V}, \quad$$ $$E^\ominus_{\text{Ag}^{+}/\text{Ag}} = +0.80\,\text{V}, \quad$$ $$\frac{2.303RT}{F} = 0.059\,\text{V}$$
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 58
Given below are two statements:
$$\textbf{Statement I :}$$ The number of pairs among $$[\text{Al}_2\text{O}_3, \text{Cr}_2\text{O}_3]$$, $$[\text{Cl}_2\text{O}_7, \text{Mn}_2\text{O}_7]$$, $$[\text{Na}_2\text{O}, \text{V}_2\text{O}_3]$$ and $$[\text{CO}, \text{N}_2\text{O}]$$ that contain oxides of same nature (acidic, basic, neutral or amphoteric) is 4.
$$\textbf{Statement II :}$$ Among $$\text{Na}_2\text{O}$$, $$\text{Al}_2\text{O}_3$$, $$\text{CO}$$ and $$\text{Cl}_2\text{O}_7$$, the most basic and acidic oxides are $$\text{Na}_2\text{O}$$ and $$\text{Cl}_2\text{O}_7$$, respectively.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 59
Given below are two statements:
$$\textbf{Statement I :}$$ Aluminium upon reaction with $$\text{NaOH}$$ forms $$[\text{Al(OH)}_6]^{3-}$$ ion.
$$\textbf{Statement II :}$$ The geometry of $$\text{ICl}_4^{-}$$, $$\text{ClO}_3^{-}$$ and $$\text{IBr}_2^{-}$$ is square planar, pyramidal and linear respectively.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 60
Given below are two statements:
$$\textbf{Statement I :}$$ Presence of large number of unpaired electrons in transition metal atoms results in higher enthalpies of their atomisation.
$$\textbf{Statement II :}$$ $$d_{xy} = d_{xz} = d_{yz} < d_{x^2-y^2} = d_{z^2}$$ and $$d_{x^2-y^2} = d_{z^2} < d_{xy} = d_{xz} = d_{yz}$$ are the d-orbital splittings in $$[\text{Fe}(\text{H}_2\text{O})_6]^{3+}$$ and $$[\text{Ni}(\text{Cl})_4]^{2-}$$ complex ions respectively.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 61
Identify the correct statements from the following
$$\textbf{A.}$$ $$[\text{Fe}(\text{C}_2\text{O}_4)_3]^{3-}$$ is the most stable complex among $$[\text{Fe}(\text{OH})_6]^{3-}$$, $$[\text{Fe}(\text{C}_2\text{O}_4)_3]^{3-}$$ and $$[\text{Fe}(\text{SCN})_6]^{3-}$$
$$\textbf{B.}$$ The stability of $$[\text{Cu}(\text{NH}_3)_4]^{2+}$$ is greater than that of $$[\text{Cu}(\text{en})_2]^{2+}$$
$$\textbf{C.}$$ The hybridization of Fe in $$\text{K}_4[\text{Fe}(\text{CN})_6]$$ is $$d^2sp^3$$
$$\textbf{D.}$$ $$[\text{Fe}(\text{NO}_2)_3\text{Cl}_3]^{3-}$$ exhibits linkage isomerism
$$\textbf{E.}$$ $$\text{NO}_2^{-}$$ and $$\text{SCN}^{-}$$ ligands are NOT ambidentate ligands
Choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 62
Match the List I with List II:

Choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 63
IUPAC name of the some alkenes are given below.
Find out the correct stability order.
$$\textbf{A.}$$ 2-Methylbut-2-ene
$$\textbf{B.}$$ $$\textit{cis}$$-But-2-ene
$$\textbf{C.}$$ 2,3-Dimethylbut-2-ene
$$\textbf{D.}$$ Prop-1-ene
Choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 64
Identify the correct IUPAC name of hydrocarbon (x) containing three primary carbons atoms and with molar mass $$72$$ g mol$$^{-1}$$ :
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 65
Complete the following reaction sequence and give the name of major product 'P'.
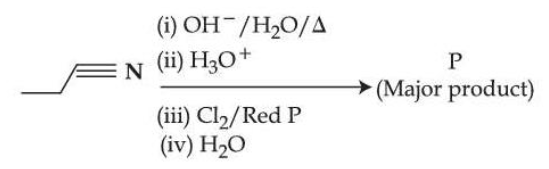
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 66
Given below are two statements:
$$\textbf{Statement I :}$$ The condensation reaction between $$\text{CH}_3-\text{CH}=\text{O}$$ and
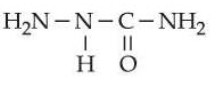
under optimum pH will produce
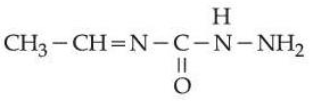
$$\textbf{Statement II :}$$ The molecule
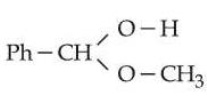
will generate $$\text{Ph}-\text{CH}=\text{O}$$ in the presence of dilute acid.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 67
Given below are two statements:
$$\textbf{Statement I :}$$ Heating benzamide with bromine in an ethanolic solution of sodium hydroxide will give benzylamine.
$$\textbf{Statement II :}$$ Nitration of aniline with $$\text{HNO}_3/\text{H}_2\text{SO}_4$$ at $$288\,\text{K}$$ produces $$m$$-nitroaniline in higher amount than $$o$$-nitroaniline (pH adjusted).
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 68
Identify the incorrect statements about tertiary structure of proteins?
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 69
Given below are two statements :
Statement I:
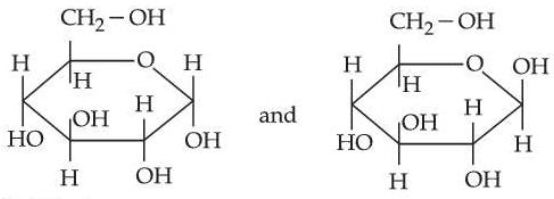
are two anomers of D-(+)-glucose.
Statement II: the Open chain forms of D-glucose and D-fructose contain three similar chiral carbons at C₃, C₄, and C₅.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 70
A paper is dipped in a dil. $$\text{H}_2\text{SO}_4$$ solution of 'X' upon treatment with $$\text{SO}_2$$ gas turns into green.The compound 'X' is :
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 71
The total number of unpaired electrons present in the $$d^3$$, $$d^4$$ (low spin), $$d^5$$ (high spin), $$d^6$$ (high spin), and $$d^7$$ (low spin) octahedral complexes systems is :
789
456
123
0.-
Clear All
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 72
$$\text{RMgI}$$ when treated with ice cold water liberated a gas which occupied $$1.4\,\text{dm}^3/\text{g}$$ at STP. The gas produced is further reacted with iodine in presence of $$\text{HIO}_3$$ to give compound (X). Compound (X) in presence of Na and dry ether produced compound (Y). Molar mass of compound (Y) is $$\underline{\hspace{2cm}}$$ $$\text{g mol}^{-1}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 73
20 g hemoglobin in a 1 L aqueous solution (A) at 300 K is separated from pure water by a semi-permeable membrane. At equilibrium, the height of solution in a tube dipped in solution (A) is found to be $$80.0 mm$$ higher than the tube dipped in water.
The molar mass of hemoglobin is ______ $$kg mol^{-1}$$. (Nearest integer)
(Given: $$g = 10 \, m \, s^{-2}$$, $$R = 8.3 \, kPa \, dm^{3} \, K^{-1} \, mol^{-1}$$, density of solution = $$1000 \, kg \, m^{-3}$$)
789
456
123
0.-
Clear All
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 74
At 298 K, the molar conductivity of an $$x\%$$ (w/w) MX solution is 123.5 S cm$$^{2} mol^{-1}$$. The conductance of same solution is $$1.9 \times 10^{-3} S$$. The value of x is ______ $$\times 10^{-2}$$.
(Given: Cell constant = 1.3 cm$$^{-1}$$, molar mass of MX = 75 $$g mol^{-1}$$, density of aqueous solution of $$MX$$ at 298 K is 1.0 $$g mL^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Mains 5th April Shift 2 2026 - Chemistry - Question 75
For the reaction $$A \to p$$ at $$T K$$, the half life ($$t_{1/2}$$) is plotted as a function of initial concentration $$[A]_o$$ of $$A$$ as give below.
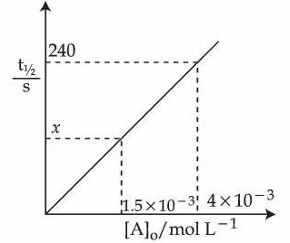
The value of $$x$$ in the given figure is ______ s (Nearest integer)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




