NTA JEE Mains 5th April Shift 1 2026 - Chemistry
For the following questions answer them individually
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 51
How many grams of residue is obtained by heating 2.76 g of silver carbonate? (Given: Molar mass of C, O and Ag are 12, 16 and 108 g mol$$^{-1}$$ respectively)
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 52
Arrange the following atomic orbitals of multi electron atoms in order of increasing energy.
A. $$n = 3, l = 2, m = +1$$
B. $$n = 4, l = 0, m = 0$$
C. $$n = 6, l = 1, m = 0$$
D. $$n = 5, l = 1, m = +1$$
E. $$n = 2, l = 1, m = +1$$
Choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 53
Identify the correct statements from the following:
A. Heisenberg uncertainty principle is applicable to electrons.
B. The size of $$2p_x$$ orbital is less than the size of $$3p_x$$ orbital.
C. The energy of 2s orbital of H atom is equal to the energy of 2s orbital of Li.
D. The electronic configuration of Cr is [Ar] $$3d^5 4s^1$$
Choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 54
What is the mole fraction of water in 10% by weight (w/w) of aqueous urea solution? [Given: Molar mass of H, O, C and N are 1, 16, 12 and 14 g mol$$^{-1}$$ respectively.]
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 55
$$M_3A_2$$ is a sparingly soluble salt of molar mass $$y$$ g mol$$^{-1}$$ and solubility $$x$$ g L$$^{-1}$$. The ratio of the molar concentration of the anion ($$A^{3-}$$) to the solubility product of the salt is
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 56
Arrange the following resultant mixtures in increasing order of their pH values
A. 10 mL 0.2 M Ca(OH)$$_2$$ + 25 mL 0.1 M HCl
B. 10 mL 0.01 M H$$_2$$SO$$_4$$ + 10 mL 0.01 M Ca(OH)$$_2$$
C. 10 mL 0.1 M H$$_2$$SO$$_4$$ + 10 mL 0.1 M KOH
Choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 57
First order gas phase reaction
$$A \to B + C$$
$$p_i$$ = initial pressure of gas A, $$p_t$$ = total pressure of the reaction mixture at time $$t$$
Expression of rate constant (k) is
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 58
Given below are two statements:
Statement I: The correct order of electronegativity of fluorine, oxygen and nitrogen is $$F > O > N$$.
Statement II: The oxidation state of oxygen in $$OF_2$$ is +2 and in $$Na_2O$$ is $$-2$$.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 59
Correct statements from the following are:
A. Nitrogen in oxidation states from +1 to +4 disproportionates in acid medium.
B. Nitrogen has the ability to form d$$\pi$$ - p$$\pi$$ multiple bonds with itself and other elements with small size and high electronegativity.
C. N-N single bond is stronger than P-P single bond.
D. Nitrogen has highest density in its group due to small size.
E. The maximum covalency of nitrogen is four since it has only four valence orbitals for bonding.
Choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 60
Which of the following is NOT a physical or chemical characteristics of interstitial compounds?
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 61
The correct statements about metal carbonyls are
A. The metal-carbon bonds in metal carbonyls possess both $$\sigma$$ and $$\pi$$-character.
B. Due to synergic bonding interactions between metal and CO ligand, the metal-carbon bond becomes weak.
C. The metal-carbon $$\sigma$$ bond is formed by the donation of lone pair of electrons on the carbonyl carbon into a vacant orbital of metal.
D. The metal-carbon $$\pi$$ bond is formed by the donation of electrons from filled d-orbital of metal into vacant $$\pi^*$$ orbital of CO.
Choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 62
Given below are two statements:
Statement I: Each electron in $$e_g$$ orbitals destabilizes the orbitals by $$+0.6 \Delta_o$$ and each electron in the $$t_{2g}$$ orbitals stabilizes the orbitals by $$-0.4 \Delta_o$$ in an octahedral field on the basis of crystal field theory.
Statement II: All the d-orbitals of the transition metals have the same energy in their free atomic state but when a complex is formed the ligands destroy the degeneracy of these orbitals on the basis of crystal field theory.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 63
Given below are two statements:
Statement I: On the basis of inductive effect, the order of stability of alkyl carbanions is $$CH_3^- > CH_3CH_2^- > (CH_3)_2CH^- > (CH_3)_3C^-$$.
Statement II: Allyl and benzyl carbanions are more stabilised by inductive effect and not by resonance effect.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 64
"P" is a hydrocarbon of molecular formula: $$C_8H_{14}$$. On ozonolysis, "P" forms "Q". "Q" on treatment with alkali under reflux condition produces "R", which on treatment with $$I_2$$/NaOH gives a yellow precipitate. Acidification of the solution gives "S". The structure of "S" is given below:-
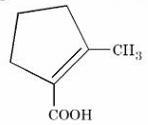
The correct structure of "P" is
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 65
For the following Friedel Craft's alkylation reaction, which of the statements are correct?
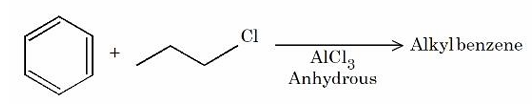
A. Major product is n-propyl benzene.
B. iso-propyl carbocation intermediate is also generated.
C. Multiple substitution is inevitable.
D. Introducing electron-donating substituent on benzene will not produce any alkyl benzene.
Choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 66
Benzyl isocyanide can be obtained from:
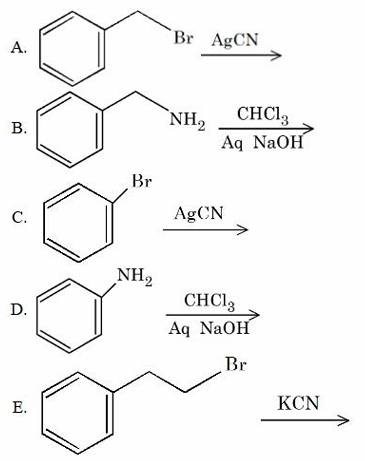
Choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 67
Consider compounds A, B and C with following structural formulae
A = $$CH_3 - CH_2 - CH_2 - CH_2 - CH_2 - OH$$
B = $$CH_2 = CH - CH_2 - CH_2 - CH_3$$
C = $$HO - CH_2 - CH_2 - CH(OH) - CH_3$$
For the conversion of B from A, reagent (D) required is _______ and structural formula of product (E) obtained when C undergoes same reaction using excess reagent (D) is _______.
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 68
Identify the incorrect statements.
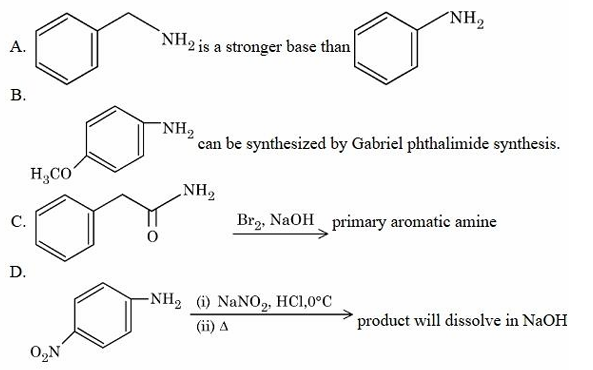
Choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 69
Identify the correct statements.
A. Glucose exists in two anomeric forms.
B. Anomers of glucose differ in configuration at C-1 in cyclic hemiacetal structure.
C. Melting point of $$\alpha$$-anomer of glucose is greater than $$\beta$$-anomer.
D. Specific rotation of $$\alpha$$-anomer is +19° while for $$\beta$$-anomer is +112°.
E. $$\alpha$$ and $$\beta$$-anomers of glucose are prepared by crystallization of saturated glucose solution at 303 K and 371 K respectively.
Choose the correct answer from the options given below:
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 70
Given below are two statements:
Statement I: Sodium dichromate and potassium dichromate are classified as primary standards in titrimetric analysis.
Statement II: Phenolphthalein is a weak base, therefore it dissociates in acidic medium.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 71
Consider the following species:
$$BrF_5$$, $$XeF_5^-$$, $$BF_4^-$$, $$ICl_4^-$$, $$XeF_4$$, $$SF_4$$, $$NH_4^+$$, $$ClF_3$$, $$XeF_2$$, $$ICl_2^-$$
Number of species having $$sp^3d$$ hybridized central atom is _______.
789
456
123
0.-
Clear All
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 72
In an estimation of sulphur by Carius method 0.2 g of the substance gave 0.6 g of BaSO$$_4$$. The percentage of sulphur in the substance is _______%. (Given molar mass in g mol$$^{-1}$$ S: 32, BaSO$$_4$$: 231)
789
456
123
0.-
Clear All
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 73
One mole of phenol is treated with dilute HNO$$_3$$ at 298 K to give a mixture of products. The mixture is separated by steam distillation. The steam volatile compound (X) is separated. The increase in percentage of oxygen in (X) with respect to phenol is _______ $$\times 10^{-1}$$ %. (Given molar mass in g mol$$^{-1}$$ H:1, C:12, N:14, O:16)
789
456
123
0.-
Clear All
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 74
The values of pressure equilibrium constant recorded at different temperatures for the following equilibrium reaction have been given below
$$A(g) \rightleftharpoons B(g) + C(g)$$
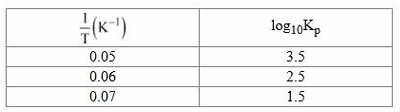
The magnitude of $$\frac{\Delta H^\circ}{R}$$ calculated from the above data is _______. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 5th April Shift 1 2026 - Chemistry - Question 75
If the half life of a first order reaction is 6.93 minutes then the time required for completion of 99% of the reaction will be _______ minutes. (Given: log 2 = 0.3010)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




