NTA JEE Mains 4th April Shift 2 2026 - Chemistry
For the following questions answer them individually
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 51
The correct order of total number of atoms in: (A) 2 moles of cyclohexane, (B) 684 g of sucrose, (C) 90.8 L of dihydrogen at STP, is :
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 52
The species having identical radii according to the Bohr's theory are:
A. H (first orbit)
B. He$$^+$$ (first orbit)
C. He$$^+$$ (Second orbit)
D. Li$$^{2+}$$ (first orbit)
E. Be$$^{3+}$$ (Second orbit)
Choose the correct answer from the options given below:
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 53
Which of the following pictorial diagram most correctly represents the $$\pi^*$$ ($$\pi$$ - antibonding) molecular orbital between two atoms if the internuclear axis is taken to be in the z-direction $$(\xrightarrow{z-axis})$$?
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 54
At 27 °C, 0.1 M, 1 L K$$_4$$[Fe(CN)$$_6$$] aqueous solution and 0.1 M, 1 L FeCl$$_3$$ aqueous solution are placed in a container separated by a semi permeable membrane AB. Assume complete dissociation of both the solutes. Which of the following statement is correct?
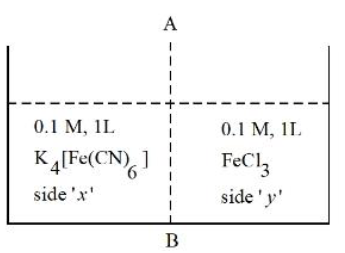
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 55
20 mL of a solution of acetic acid required 28.4 mL of 0.1 M NaOH for its neutralization. A solution (X) was prepared by mixing 20 mL of the above acetic acid and 14.2 mL of 0.1 M NaOH solution. What is the pH of the solution (X)? (pK$$_a$$ value of acetic acid is 4.75).
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 56
Match the List I with List II:
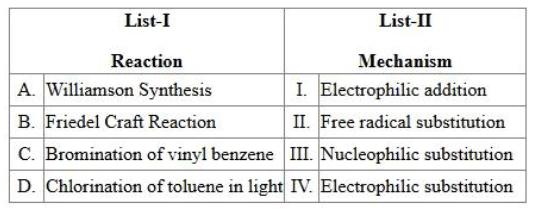
Choose the correct answer from the options given below
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 57
The 1$$^{st}$$ ionization enthalpy for Mg is +737 kJ/mol. The most probable estimated value of the 2$$^{nd}$$ ionization enthalpy of Mg is ______.
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 58
The electronegativity of a group 13 element 'E' is same as that of Ge (on Pauling scale and upto one decimal point). The CORRECT statements about E$$^{3+}$$ are
A. It can act as a reducing agent.
B. It can act as an oxidizing agent.
C. E$$^{3+}$$ is more stable than E$$^+$$.
D. The standard electrode potential value for E$$^{3+}$$/E is positive.
choose the correct answer from the options given below
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 59
Pairs of elements with the same number of electrons in their respective 4f orbital are
[Atomic number. Eu-63, Gd-64, Dy-66, Ho-67, Tm-69, Yb-70, Lu-71, Hf-72]
A. (Eu and Gd)
B. (Dy and Ho)
C. (Yb and Hf)
D. (Lu and Tm)
choose the correct answer from the options given below
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 60
Consider the metal complexes $$[Ni(en)_3]^{2+}$$ (A), $$[NiCl_4]^{2-}$$ (B) and $$[Ni(NH_3)_6]^{2+}$$ (C). Choose the CORRECT option by considering the number of unpaired electrons present in (A), (B) and (C) respectively and the order of frequency of absorption
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 61
Consider the following molecules/species:
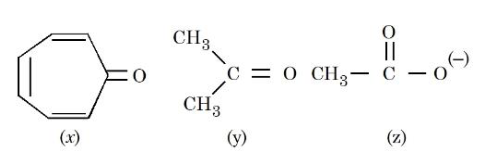
The correct order of carbon-oxygen double bond length is:
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 62
Consider $$|x|$$ is the difference in oxidation states of Mn in highest manganese fluoride and highest manganese oxide. The ions with $$|x|$$ number of unpaired electrons from the following are:
A. Sc$$^{3+}$$
B. Zn$$^{2+}$$
C. V$$^{2+}$$
D. Fe$$^{2+}$$
E. Co$$^{2+}$$
choose the correct answer from the options given below :
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 63
Consider the given graph showing variation of reactant concentration with time. Three different reactions were started with identical initial concentration of reactants. Which of the following statement is correct?
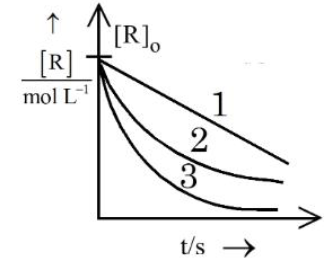
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 64
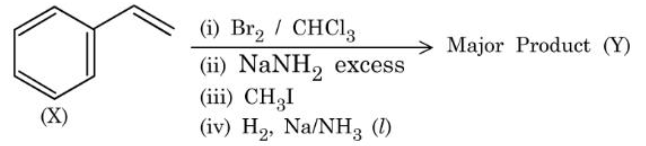
Compound (X) is subjected to the sequence of reactions as shown above:
Molar mass of the major product (Y) formed is ______ g mol$$^{-1}$$.
(Given molar mass in g mol$$^{-1}$$ C:12, H: 1, O: 16)
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 65
The following structures are
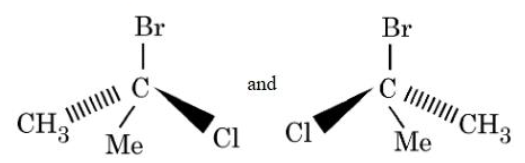
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 66
The descending order of acidity among the following compounds is:
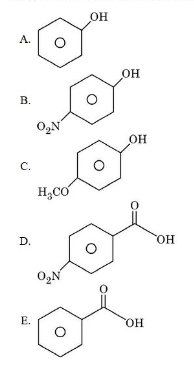
choose the correct answer from the options given below:
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 67
The strongest conjugate acid will result from:
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 68
A D-aldotetrose on oxidation with concentrated HNO$$_3$$ resulted in optically inactive dicarboxylic acid. The structure of the D-aldotetrose is:
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 69
Among Fe$$^{3+}$$, Pb$$^{2+}$$, Cu$$^{2+}$$ and Mn$$^{2+}$$, identify the one that gets precipitated out while passing H$$_2$$S in presence of NH$$_4$$OH as group reagent. The highest possible oxidation state of the corresponding metal is
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 70
Match the List I with List II:
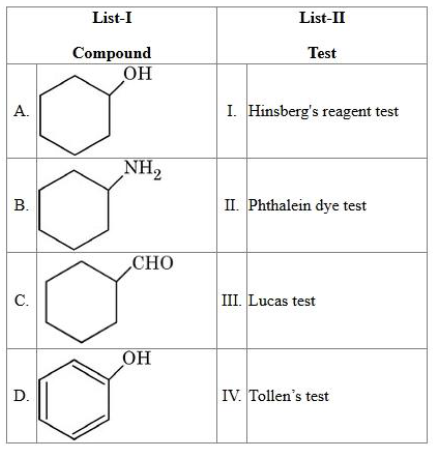
choose the correct answer from the options given below
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 71
If 3.365 g of ethanol (l) is burnt completely in a bomb calorimeter at 298.15 K, the heat produced is 99.472 kJ. The $$|\Delta H_f°|$$ of ethanol at 298.15 K is _________ $$\times 10^2$$ kJ mol$$^{-1}$$. (Nearest integer)
Given: Standard enthalpy for combustion of graphite = $$-393.5$$ kJ mol$$^{-1}$$
Standard enthalpy of formation of water (l) = $$-285.8$$ kJ mol$$^{-1}$$
Molar mass in g mol$$^{-1}$$ of C, H, O are 12, 1 and 16 respectively
789
456
123
0.-
Clear All
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 72
For the following reaction at 50 °C and at 2 atm pressure,
$$2N_2O_5(g) \rightleftharpoons 2N_2O_4(g) + O_2(g)$$
N$$_2$$O$$_5$$ is 50% dissociated.
The magnitude of standard free energy change at this temperature is $$x$$.
$$x$$ = _________ J mol$$^{-1}$$ [Nearest integer].
Given: $$R = 8.314$$ J mol$$^{-1}$$ K$$^{-1}$$, $$\log 2 = 0.30$$, $$\log 3 = 0.48$$, $$\ln 10 = 2.303$$, °C + 273 = K
789
456
123
0.-
Clear All
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 73
An electrochemical cell, consist of the following two redox couples, M$$^{x+}$$(aq)/M(s) [$$E_{red}^{\Theta} = +0.15$$ V] and Fe$$^{3+}$$(aq)/Fe(s) [$$E_{red}^{\Theta} = -0.036$$ V]. The cell EMF (E$$_{cell}$$) is recorded to be 0.2057 V. If the reaction quotient of the electrochemical reaction is found to be $$10^{-2}$$, then the value of $$x$$ is __________. (Nearest integer)
[Given: M is a p-block metal and $$\frac{2.303RT}{F} = 0.059$$ V]
789
456
123
0.-
Clear All
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 74
For a first order reaction A $$\to$$ B
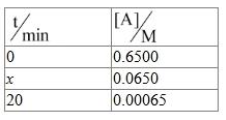
$$x$$ = _________ min. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 4th April Shift 2 2026 - Chemistry - Question 75
In sulphur estimation, $$2.0 \times 10^{-3}$$ mol of an organic compound (X) (molar mass 76 g mol$$^{-1}$$) gave 0.4813 g of barium sulphate (molar mass 233 g mol$$^{-1}$$). The percentage of sulphur in the compound (X) is _________ $$\times 10^{-1}$$ % (Nearest integer)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




