NTA JEE Mains 4th April Shift 1 2026
For the following questions answer them individually
NTA JEE Mains 4th April Shift 1 2026 - Question 71
According to Lewis theory, the total number of $$\sigma$$ bond-pairs and lone pair of electrons around the central atom of $$\text{XeO}_6^{4-}$$ ion is _____.
789
456
123
0.-
Clear All
NTA JEE Mains 4th April Shift 1 2026 - Question 72
Consider the following sequence of reactions to give the major product $$(X)$$:
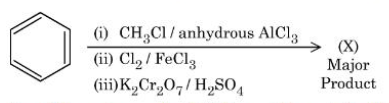
$$P$$ g of the major product $$(X)$$ formed is reacted with $$\text{NaHCO}_3$$ solution to liberate a gas which occupied 11.2 dm³ at STP. $$P$$ = _____ g.
( Given molar mass in g $$mol^{-1} H:1,C:12,O:16,Cl:35.5$$)
789
456
123
0.-
Clear All
NTA JEE Mains 4th April Shift 1 2026 - Question 73
2.0 g of a bromo hydrocarbon $$(X)$$ was subjected to Carius analysis, gave 3.36 g of AgBr. The percentage of carbon in the compound $$(X)$$ is 26.7%. Total number of carbon atoms in the empirical formula for compound $$(X)$$ is _____.
( Given molar mass in g $$mol^{-1}H:1,C:12,O:80,Cl:108$$)
789
456
123
0.-
Clear All
NTA JEE Mains 4th April Shift 1 2026 - Question 74
The pH of a solution obtained by mixing 5 mL of 0.1 M $$\text{NH}_4\text{OH}$$ solution with 250 mL of 0.1 M $$\text{NH}_4\text{Cl}$$ solution is _____ $$\times 10^{-2}$$. (Nearest integer)
Given: $$\text{pK}_b(\text{NH}_4\text{OH}) = 4.74$$, $$\log 2 = 0.30$$, $$\log 3 = 0.48$$, $$\log 5 = 0.70$$
789
456
123
0.-
Clear All
NTA JEE Mains 4th April Shift 1 2026 - Question 75
A non-volatile, non-electrolyte solid solute when dissolved in 40 g of a solvent, the vapour pressure of the solvent decreased from 760 mm Hg to 750 mm Hg. If the same solution boils at 320 K, then the number of moles of the solvent present in the solution is _____. (Nearest integer)
[Given: boiling point of the pure solvent = 319.5 K, $$K_b$$ of the solvent = 0.3 K kg mol$$^{-1}$$]
789
456
123
0.-
Clear All


