NTA JEE Mains 4th April Shift 1 2026 - Chemistry
For the following questions answer them individually
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 51
Number of moles and number of molecules in 1.4187 L of $$\text{SO}_2$$ at STP respectively are :
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 52
What is the ratio of wave number of first line (lowest energy line) of Balmer series of H atomic spectrum to first line of its Brackett series?
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 53
Which of the following is correct set of 4 quantum numbers of 19th electron in Chromium (Atomic number = 24) in accordance with Aufbau principle?
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 54
Given below are two statements:
Statement I: For an ideal gas, heat capacity at constant volume is always greater than the heat capacity at constant pressure.
Statement II: In a constant volume process, no work is produced and all the heat withdrawn goes into the chaotic motion and is reflected by a temperature increase of the ideal gas.
In the light of the above statement, choose the correct answer from the options given below.
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 55
At $$T$$(K), the equilibrium constant of $$A_2(g) + B_2(g) \rightleftharpoons C(g)$$ is $$2.7 \times 10^{-5}$$. What is the equilibrium constant for $$\frac{1}{3}A_2(g) + \frac{1}{3}B_2(g) \rightleftharpoons \frac{1}{3}C(g)$$ at the same temperature?
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 56
In order to oxidise a mixture of 1 mole each of $$\text{FeC}_2\text{O}_4$$, $$\text{Fe}_2(\text{C}_2\text{O}_4)_3$$, $$\text{FeSO}_4$$ and $$\text{Fe}_2(\text{SO}_4)_3$$ in acidic medium, the number of moles of $$\text{KMnO}_4$$ required is :
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 57
Consider the first order reaction $$R \to P$$. The fraction of molecules decomposed in the given first order reaction can be expressed as :
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 58
A monoatomic anion $$(A^-)$$ has 45 neutrons and 36 electrons. Atomic mass, group in the periodic table and physical state at room temperature of the element $$(A)$$ respectively are :
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 59
Given below are two statements:
Statement I: The covalency of oxygen is generally two but it can exceed upto four. The oxidation state of oxygen in $$\text{SO}_2$$ is $$-2$$ and in $$\text{OF}_2$$ it is $$+2$$.
Statement II: The anomalous behaviour of oxygen when compared to the other elements of group 16 is due to its small size and high electronegativity.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 60
The correct statements among the following are:
A. Mo(VI) and W(VI) are less stable than Cr(VI).
B. Ce$$^{4+}$$ and Tb$$^{4+}$$ are oxidant while Eu$$^{2+}$$ and Yb$$^{2+}$$ are reductant.
C. Cm and Am have seven unpaired electrons.
D. Actinoid contraction is greater from element to element than lanthanoid contraction.
choose the correct answer from the options given below:
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 61
Correct statements from the following are:
A. Potassium dichromate is an oxidising agent and it oxidises $$\text{FeSO}_4$$ to $$\text{Fe}_2(\text{SO}_4)_3$$ in acidic medium.
B. Sodium dichromate can be used as primary standard in volumetric estimation.
C. $$\text{CrO}_4^{2-}$$ and $$\text{Cr}_2\text{O}_7^{2-}$$ are interconvertible in aqueous solution by varying the pH of the solution.
D. Cr-O-Cr bond angle in $$\text{Cr}_2\text{O}_7^{2-}$$ is 126°.
choose the correct answer from the options given below
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 62
Match the LIST-I with LIST-II.
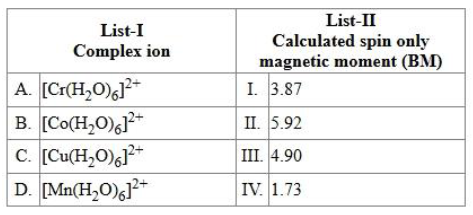
choose the correct answer from the options given below
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 63
Increasing order of electron withdrawing power of following functional groups is:
a. $$-\text{CN}$$, b. $$-\text{COOH}$$, c. $$-\text{NO}_2$$, d. $$-\text{I}$$
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 64
An alkene $$(X)$$ on ozonolysis followed by reduction gives following products:
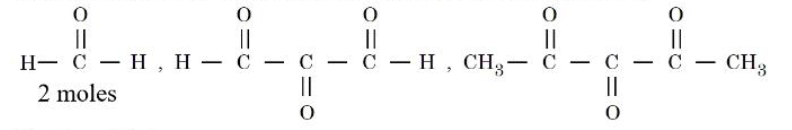
The alkene $$(X)$$ is :
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 65
Match List-I with List-II.
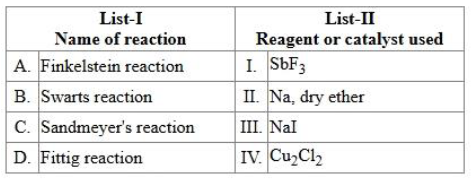
Choose the correct answer from the options given below :
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 66
Amongst the following, the total number of compounds soluble in aqueous NaOH at room temperature is:
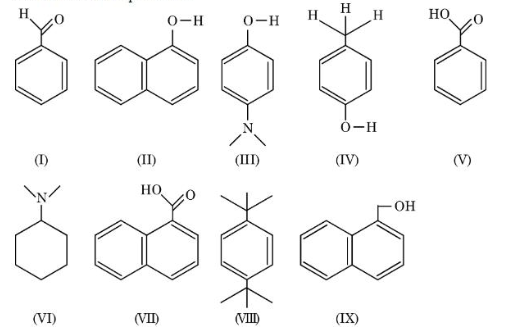
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 67
Product C of the following reaction sequence will be:
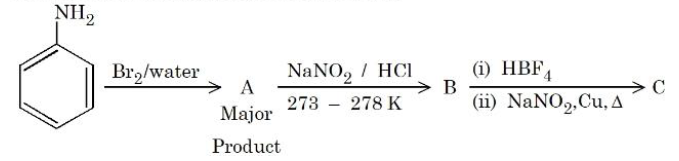
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 68
Given below are two statements:
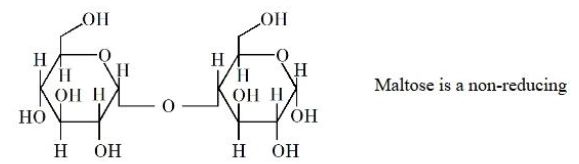
Statement I: The structure of Maltose is given below. Maltose is a non-reducing sugar.
Statement II: The structure of Lactose is given below. Lactose is a reducing sugar.
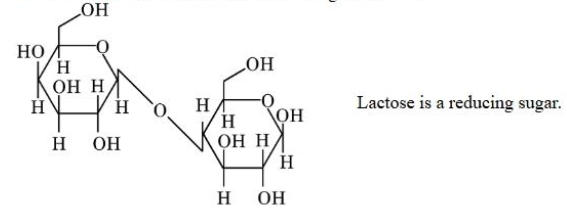
in the light of the above statements, Choose the correct answer from the options given below :
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 69
Match List-I with List-II.
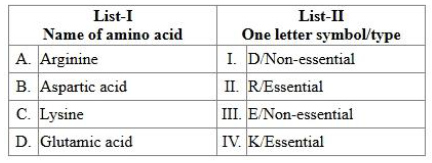
Choose the correct answer from the options given below :
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 70
Identify the colour of compound 'X' in the sequence of the reaction:
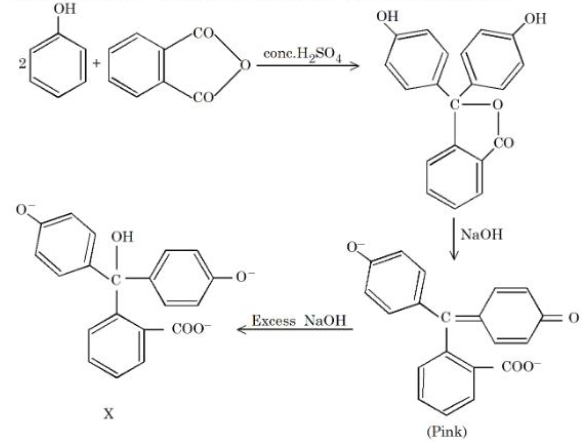
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 71
According to Lewis theory, the total number of $$\sigma$$ bond-pairs and lone pair of electrons around the central atom of $$\text{XeO}_6^{4-}$$ ion is _____.
789
456
123
0.-
Clear All
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 72
Consider the following sequence of reactions to give the major product $$(X)$$:
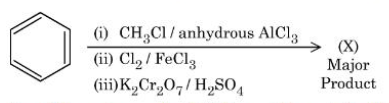
$$P$$ g of the major product $$(X)$$ formed is reacted with $$\text{NaHCO}_3$$ solution to liberate a gas which occupied 11.2 dm³ at STP. $$P$$ = _____ g.
( Given molar mass in g $$mol^{-1} H:1,C:12,O:16,Cl:35.5$$)
789
456
123
0.-
Clear All
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 73
2.0 g of a bromo hydrocarbon $$(X)$$ was subjected to Carius analysis, gave 3.36 g of AgBr. The percentage of carbon in the compound $$(X)$$ is 26.7%. Total number of carbon atoms in the empirical formula for compound $$(X)$$ is _____.
( Given molar mass in g $$mol^{-1}H:1,C:12,O:80,Cl:108$$)
789
456
123
0.-
Clear All
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 74
The pH of a solution obtained by mixing 5 mL of 0.1 M $$\text{NH}_4\text{OH}$$ solution with 250 mL of 0.1 M $$\text{NH}_4\text{Cl}$$ solution is _____ $$\times 10^{-2}$$. (Nearest integer)
Given: $$\text{pK}_b(\text{NH}_4\text{OH}) = 4.74$$, $$\log 2 = 0.30$$, $$\log 3 = 0.48$$, $$\log 5 = 0.70$$
789
456
123
0.-
Clear All
NTA JEE Mains 4th April Shift 1 2026 - Chemistry - Question 75
A non-volatile, non-electrolyte solid solute when dissolved in 40 g of a solvent, the vapour pressure of the solvent decreased from 760 mm Hg to 750 mm Hg. If the same solution boils at 320 K, then the number of moles of the solvent present in the solution is _____. (Nearest integer)
[Given: boiling point of the pure solvent = 319.5 K, $$K_b$$ of the solvent = 0.3 K kg mol$$^{-1}$$]
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




