NTA JEE Mains 31st Jan 2024 Shift 1
For the following questions answer them individually
NTA JEE Mains 31st Jan 2024 Shift 1 - Question 51
Number of moles of methane required to produce $$22$$ g $$CO_{2(g)}$$ after combustion is $$x \times 10^{-2}$$ moles. The value of $$x$$ is
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 1 - Question 52
The ionization energy of sodium in $$kJ \text{ mol}^{-1}$$, if electromagnetic radiation of wavelength $$242$$ nm is just sufficient to ionize sodium atom is ______. (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 1 - Question 53
The number of species from the following in which the central atom uses $$sp^3$$ hybrid orbitals in its bonding is _________. $$NH_3, SO_2, SiO_2, BeCl_2, CO_2, H_2O, CH_4, BF_3$$
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 1 - Question 54
Consider the following reaction at 298 K. $$\frac{3}{2}O_{2(g)} \rightleftharpoons O_{3(g)}$$, $$K_p = 2.47 \times 10^{-29}$$. $$\Delta_r G^0$$ for the reaction is _________ kJ. (Given $$R = 8.314 \text{ JK}^{-1}\text{mol}^{-1}$$) Round off your answer to the nearest integer.
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 1 - Question 55
Number of alkanes obtained on electrolysis of a mixture of $$CH_3COONa$$ and $$C_2H_5COONa$$ is _____.
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 1 - Question 56
One Faraday of electricity liberates $$x \times 10^{-1}$$ gram atom of copper from copper sulphate, $$x$$ is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 1 - Question 57
The 'Spin only' Magnetic moment for $$[Ni(NH_3)_6]^{2+}$$ is ______ $$\times 10^{-1}$$ BM. (given = Atomic number of Ni : 28) Round off your answer to the nearest integer.
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 1 - Question 58
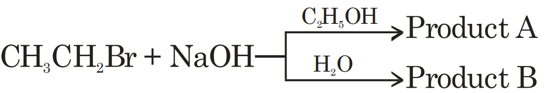
The total number of hydrogen atoms in product A and product B is __________.
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 1 - Question 59
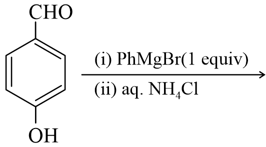
The number of hydroxyl groups present in the product $$P$$ is __________.
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 1 - Question 60
Molar mass of the salt from $$NaBr, NaNO_3, KI$$ and $$CaF_2$$ which does not evolve coloured vapours on heating with concentrated $$H_2SO_4$$ is ____ g mol$$^{-1}$$. (Molar mass in g mol$$^{-1}$$: Na: 23, N: 14, K: 39, O: 16, Br: 80, I: 127, F: 19, Ca: 40)
789
456
123
0.-
Clear All


