NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 51

Choose the correct answer from the options given below :
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 52
500J of energy is transferred as heat to 0.5 mol of Argon gas at 298 K and 1.00 atm. The final temperature and the change in internal energy respectively are: Given : $$R = 8.3 J K^{-1}mol^{-1}$$
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 53
At temperature $$T$$, compound $$AB_{2(g)}$$ dissociates as $$AB_{2} \rightleftharpoons AB_{(g)}+\frac{1}{2}B_{2(g)}$$ having degree of dissociation $$x$$ (small compared to unity). The correct expression for $$x$$ in terms of $$K_{p}$$ and p is
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 54
An element 'E'has the ionisation enthalpy value of $$374 kJ mol^{-1}$$.'E'reacts with elements A, B, C and D with electron gain enthalpy values of −328, −349, −325 and $$-295 kJ mol^{-1}$$, respectively. The correct order of the products EA, EB, EC and ED in terms of ionic character is :
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 55
Total number of nucleophiles from the following is :
$$NH_{3},PhSH,(H_{3}C)_{2}S,H_{2}C=CH_{2},\ominus\\O H,H_{3}O^{\oplus},(CH_{3})_{2}CO,\rightleftharpoons NCH_{3}$$
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 56
The steam volatile compounds among the following are :
(A)
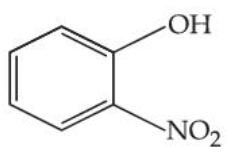
(B)
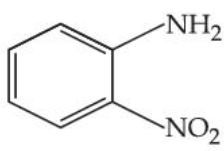
(C)
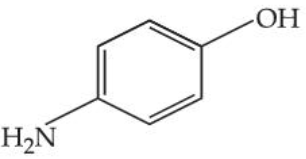
(D)
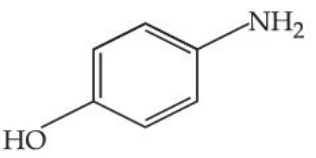
Choose the correct answer from the options given below :
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 57
Given below are two statements : Statement (I): The radii of isoelectronic species increases in the order. $$Mg^{2+} < Na^{+} < F^{-} < O^{2-}$$ Statement (II): The magnitude of electron gain enthalpy of halogen decreases in the order. Cl > F > Br > I In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 58
Match List - I with List - II.

choose the correct answer from the options given below :
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 59
The molar conductivity of a weak electrolyte when plotted against the square root of its concentration, which of the following is expected to be observed ?
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 60
The standard reduction potential values of some of the p-block ions are given below. Predict the one with the strongest oxidising capacity.
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 61
The product (P) formed in the following reaction is :
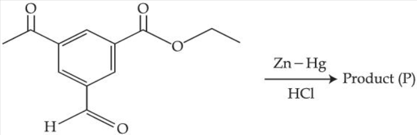
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 62
If $$a_{\circ}$$ is denoted as the Bohr radius of hydrogen atom, then what is the de-Broglie wavelength $$(\lambda)$$ of the electron present in the second orbit of hydrogen atom? [n : any integer]
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 63
Match List - I with List - II.
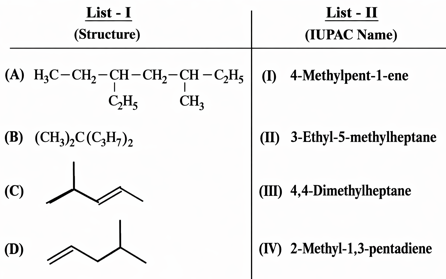
Choose the correct answer from the options given below :
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 64
The reaction $$A_{2}+B_{2}\rightarrow 2AB$$ follows the mechanism
$$A_{2}k_{1}A+A(fast)\\ \text{ }{_{k_{-1}}}\\ \\A+B_{2}\xrightarrow{k_{2}}AB+B(slow)\\A+B \rightarrow AB(fast)$$
The overall order of the reaction is :
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 65
1.24 g of $$AX_{2}$$ (molar mass 124 g $$mol^{-1}$$) is dissolved in 1 kg of water to form a solution with boiling point of $$100.0156^{\circ}C$$, while $$25.4g^{\circ}$$ of $$AY_{2}$$ (molar mass 250 g $$mol^{-1}$$) in 2 kg of water constitutes a solution with a boiling point of $$100.0260^{\circ}C.K_{b}(H_{2}O)=0.52 K$$ kg $$mol^{-1}$$ Which of the following is correct ?
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 66
Choose the correct statements. (A) Weight of a substance is the amount of matter present in it. (B) Mass is the force exerted by gravity on an object. (C) Volume is the amount of space occupied by a substance. (D) Temperatures below $$O^{\circ}C$$ are possible in Celsius scale, but in Kelvin scale negative temperature is not possible. (E) Precision refers to the closeness of various measurements for the same quantity. Choose the correct answer from the options given below :
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 67
The correct option with order of melting points of the pairs (Mn, Fe), (Tc, Ru) and (Re, Os) is :
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 68
For a $$Mg|Mg^{2+}(aq)||Ag^{+}(aq)|Ag$$ the correct Nernst Equation is :
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 69
In The following substitution reaction :
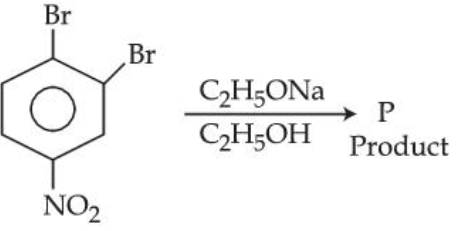
product 'P'formed is :
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 70
The correct increasing order of stability of the complexes based on $$\Delta_{\circ}$$ value is: I.$$[Mn(CN)_{6}]^{3}$$ II.$$[Co(CN)_{6}]^{4-}$$ III.$$[Fe(CN)_{6}]^{4-}$$ IV.$$[Fe(CN)_{6}]^{3-}$$
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 71
The molar mass of the water insoluble product formed from the fusion of chromite ore $$(FeCr_{2}O_{4})$$ with $$Na_{2}CO_{3}$$ in presence of $$O_{2}$$ is_______$$gmol^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 72
Given below are some nitrogen containing compounds
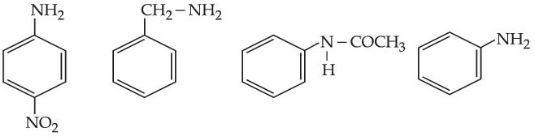
Each of them is treated with HCl separately. 1.0 g of the most basic compound will consume _______ mg of HCl. (Given molar mass in $$gmol^{-1}$$ C : 12, H : 1, O : 16, Cl : 35.5)
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 73
The sum of sigma $$(\sigma)$$ and pi$$(\pi)$$ bonds in Hex-1,3-dien-5-yne is _______.
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 74
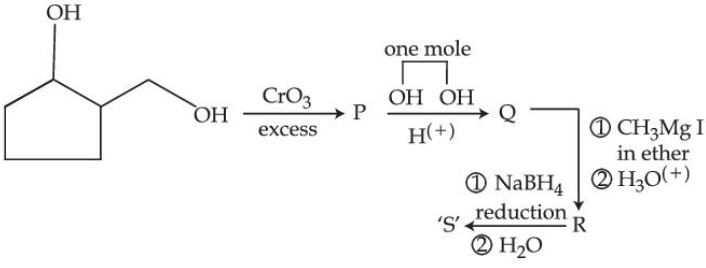
0.1 mole of compound ' S ' will weigh _______ g. (Given molar mass in $$gmol^{-1}$$ C : 12, H : 1, O : 16)
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2025 Shift 1 - Chemistry - Question 75
If $$A_{2}B$$ is 30% ionised in an aqueous solution, then the value of van't Hoff factor (i) is ______ $$\times 10^{-1}$$
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




