NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 51
Identify the conect statements:
The presence of - $$NO_{2}$$ group in benzene ring
A. activates the ring towards electrophilic substitutions.
B. deactivates the ring towards electrophilic substitutions.
C. activates the ring towards nucleophilic substitutions.
D. deactivates the ring towards nucleophilic substitutions.
choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 52
The wavelength of photon ' A' is 400 nm. The frequency of photon ' B' is $$10^{16}s^{-1}$$. The wave number of photon 'C is $$10^{4}cm^{-1}$$.The correct order of energy of these photons is :
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 53
A student performed analysis of aliphatic organic compound 'X' which on analysis gave C =61.01 % H =15.25%, N=23.74%.
This compound, on treatment with $$HNO_{2}/H_{2}O$$ produced another compound 'Y' which did not contain any nitrogen atom However, the compound 'Y' upon controlled oxidation produced another compound 'Z' that responded to iodoform test.
The structure of 'X' is :
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 54
which of the following reaction is NOT correctly represented?
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 55
Total number of alkali insoluble solid sulphonamides obtained by reaction of given amines with Hinsberg's reagent is_______.
Aniline, N-Methylaniline, Methanamine, N,N-Dimethylmethanamine, N-Methyl methanamine, Phenylmethanamine, N-propylaniline, N-phenylaniline, N,N-Dimethylaniline, Allyl amine,Isopropyl amine
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 56
The correct increasing o rd e r of spin-only magnetic moment values of the complex ions $$[MnBr_{4}]^{2-}(A),[Cu(H_{2}O)_{6}]^{2+}(B),[Ni(CN)_{4}]^{2-}(C)$$ and $$[Ni(H_{2}O)_{6}]^{2+}(D)$$ is:
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 57
Observe the following equilibrium in a 1 L flask.
$$A(g)\rightleftharpoons B(g)$$
At T(K), the equilibrium concentrations of A and B are 0.5 Mand 0.375 M respectively. 0.1 moles of A is added into the flask and heated to T(K) to establish the equilibrium again. The new equilibrium concentrations (in M) of A and B are respectively
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 58
Given below are two statements :
Statement I : The increasing order of boiling point of hydrogen halides is $$HCl < HBr < HI < HF$$.
Statement II: The increasing order of melting point of hydrogen halides is $$HCl < HBr < HF < HI$$.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 59
Structures of four disaccharides are given below. Among the given disaccharides, the non-reducing sugar is :
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 60
For the given reaction;
$$CaCO_{3}+2HCl\rightarrow CaCl_{2}+H_{2}O+CO_{2}$$
If 90 g $$CaCO_{3}$$ is added to 300 mL of HCI which contains 38.55% HCI by mass and has density 1.13 g $$mol^{-1}$$, then which of the following option is correct ?
Given molar mass of H, Cl, Ca and Oare 1, 35.5, 40 and 16 g $$mol^{-1}$$ respectively.
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 61
Match List - I with List - Il according to shape.
List - I List - II
A. $$ XeO_3 $$ I. $$ BrF_5 $$
B. $$ XeF_2 $$ II. $$ NH_3 $$
C. $$ XeO_2 F_2 $$ III. $$ [I_3]^-{} $$
D. $$ XeOF_4 $$ IV. $$ SF_4 $$
Choose the correct answer from the options given below:
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 62
The plot of $$\log_{10}^{K} VS \frac{1}{T}$$ gives a straight Line. The intercept and slope respectively are
(where K is equilibrium constant).
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 63
Consider the elements N, P, 0, S,Cl and F. The number of valence electrons present in the elements with most and least metallic character from the above list is respectively.
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 64
Consider the following aqueous solutions.
I. 2.2 g Glucose in 125 ml of solution.
II. 1.9 g Calcium chloride in 250 ml of solution.
111. 9.0 g Urea in 500 ml of solution.
IV. 20.5 g Aluminium sulphate in 750 ml of solution.
The correct increasing order of boiling point of these solutions will be:
[Given : Molar mass in g $$mol^{-1}$$: H = 1, C=12, N= 14, 0=16, Cl =35.5, Ca=40, Al=27 and S=32]
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 65
Consider the following reactions
$$\begin{aligned}\mathrm{Na_2B_4O_7} & \xrightarrow{\Delta} 2X + Y \\[6pt]\mathrm{CuSO_4} + Y &\xrightarrow{\text{Non-luminous flame}} Z + \mathrm{SO_3}\\[6pt]2Z + 2X + \mathrm{C}&\xrightarrow{\text{Luminous flame}} 2Q +\mathrm{Na_2B_4O_7} + \mathrm{CO}\end{aligned}$$
The oxidation states of Cu in Z and Q, respectively are:
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 66
Consider the following statements about manganate and permanganate ions. Identify the correct statements.
A. The geometry of both manganate and permanganate ions is tetrahedral
B. The oxidation states of Mn in manganate and permanganate are + 7 and + 6, respectively.
C. Oxidation of Mn(II) salt by peroxodisulphate gives manganate ion as the final product.
D. Manganate ion is paramagnetic and permanganate ions is diamagnetic.
E. Acidified permanganate ion reduces oxalate, nitrite and iodide ions.
Choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 67
A student has been given 0.314 g of an organic compound and asked to estimate Sulphur. During the experiment, the student has obtained 0.4813 g of barium sulphate. The percentage of sulphur present in the compound is_______.{Given Molor mass in g $$mol^{-1}$$ S: 32, $$BaSO_{4}$$ : 233)
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 68
The cyclic cations having the same number of hyperconjugalion are :
A.
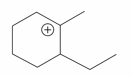
B.
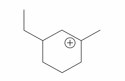
C.
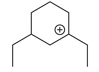
D.
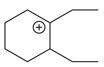
Choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 69
The correct order of acidic strength of the major products formed in the given reactions, is:
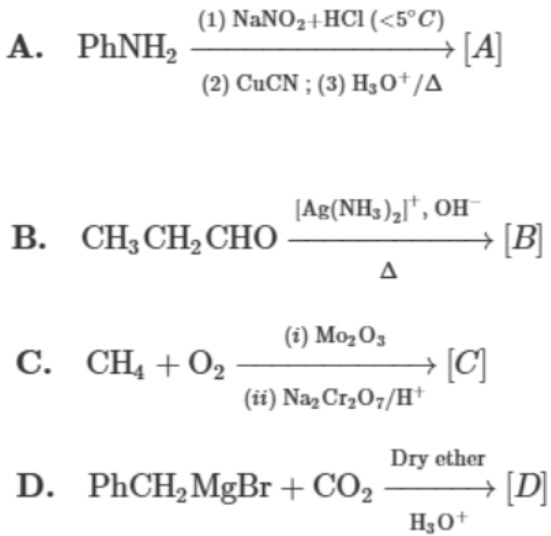
Choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 70
The reactions which produce alcohol as the product are:
$$A. CH_{4}+O_{2}\xrightarrow[\Delta]{Mo_{2}O_{3}}\\B.2CH_{3}CH_{3}+3O_{2}\xrightarrow[\Delta]{(CH_{3}COO)_{2}Mn}\\C.(CH_{3})_{3}CH\xrightarrow{KMnO_{4}}\\D.2CH_{4}+O_{2}\xrightarrow{Cu/523K/100atm.}\\E.CH_{3}-CH=CH-CH_{3}\xrightarrow{KMnO_{4}/H^{+}}$$
Choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 71
A volume of x mL of 5 M $$NaHCO_{3}$$ solution was mixed with 10 mL of 2 M $$H_{2}CO_{3}$$ solution to make an electrolytic buffer. If the same buffer was used in the following electrochemical cell to record a cell potential of 235.3 mV, then the value of x=_______ mL (nearest integer).
$$Sn(s)|Sn(OH)_{6}^{2-}(0.5 M)|HSnO_{2}^{-}(0.05 M)|OH^{-}|Bi_{2}O_{3}(s)|Bi(s)$$
Consider upto one place of decimal for intermediate calculations

789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 72
The numbei of isoelectronic species among $$Sc^{3+},Cr^{2+},Mn^{3+},Co^{3+}\text{ and }Fe^{3+}$$ is 'n'. If 'n' moles of AgCl is formed during the reaction of complex with formula $$CoCl_{3}(en)_{2}NH_{3}$$ with excess of $$AgNO_{3}$$ solution, then the number of electrons present in the $$t_{2g}$$ orbital of the complex is ________.
789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 73
For strong electrolyte $$\lambda_m$$ increases slowly with dilution and can be represented by the equation
$$\Lambda_m = \Lambda_m^\circ - A c^{1/2}$$
Molar conductivity values of the solutions of strong electrolyte AB at 18°C are given below:
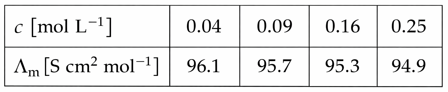
The value of constant A based on the above data [in S $$cm^{2}mol^{-1}/(mol/L)^{1/2}$$]unit is_______.
789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 74
Two positively charged particles m1 and m2 have been accelerated across the same potential difference of 200 keV as shown below.
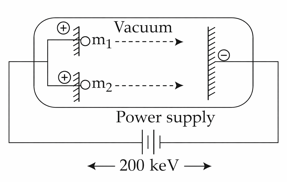
[Given mass of $$m_{1}$$ = 1amu and $$m_{2}$$ = 4amu]
The deBroglie wavelength of $$m_{1}$$ will be x times of $$m_{2}$$. The value of x is_______(nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2026 Shift 2 - Chemistry - Question 75
$$A \rightarrow B$$ (first reaction)
$$C \rightarrow D$$ (second reaction)
Consider the above two first-order reactions. The rate constant for first reaction at 500 K is double of the same at 300 K. At 500 K, 50% of the reaction becomes complete in 2 hour. The activation energy of the second reaction is half of that of first reaction. lf the rate constant at 500 K of the second reaction becomes double of the rate constant of first reaction at the same temperature; then rate constant for the second reaction at 300 K is______$$\times 10^{-1}hour^{-1}$$ (nearest integer).
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




