NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 51
Concentrated nitric acid is labelled as 75% by mass. The volume in mL of the solution which contains 30 g of nitric acid is ____. Given : Density of nitric acid solution is 1.25 g/mL .
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 52
Given below are two statements : Statement (I) :
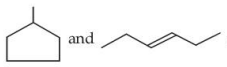
are isomeric compounds. Statement (II) :
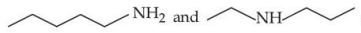
are functional group isomers.
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 53
Identify product [A],[B] and [C] in the following reaction sequence.
$$\mathrm{CH_3 - C \equiv CH \xrightarrow[\mathrm{H_2}]{\mathrm{Pd/C}} [A] \xrightarrow[\mathrm{(ii)\ Zn,\ H_2O}]{\mathrm{(i)\ O_3}} [B] + [C]}$$
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 54
The total number of compounds from below when treated with hot $$KMnO_{4}$$ giving benzoic acid is :
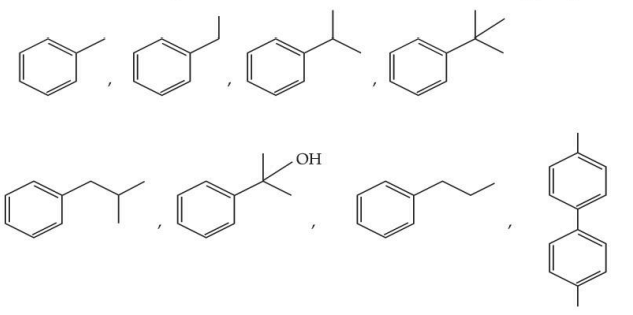
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 55
Match List - I with List - II.

Choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 56
Identify correct statements : (A) Primary amines do not give diazonium salts when treated with $$NaNO_{2}$$ in acidic condition. (B) Aliphatic and aromatic primary amines on heating with $$CHCl_{3}$$and ethanolic KOH form carbylamines. (C) Secondary and tertiary amines also give carbylamine test. (D) Benzenesulfonyl chloride is known as Hinsberg's reagent. (E) Tertiary amines reacts with benzenesulfonyl chloride very easily. Choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 57
For bacterial growth in a cell culture, growth law is very similar to the law of radioactive decay. Which of the following graphs is most suitable to represent bacterial colony growth ? Where N - Number of Bacteria at any time, $$N_{0}$$ - Initial number of Bacteria.
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 58
The amphoteric oxide among $$V_{2}O_{3}$$ and $$V_{2}O_{5}$$ , upon reaction with alkali leads to formation of an oxide anion. The oxidation state of V in the oxide anion is :
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 59
Arrange the following in increasing order of solubility product :$$\mathrm{Ca(OH)_2, \ AgBr, \ PbS, \ HgS}$$
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 60
The purification method based on the following physical transformation is :
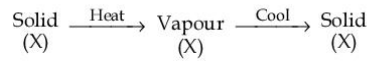
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 61
Which of the following is/are not correct with respect to energy of atomic orbitals of hydrogen atom? (A)1s < 2p < 3d < 4s (B) 1s < 2s = 2p < 3s = 3p (C)1s < 2s < 2p < 3s < 3p (D) 1s < 2s < 4s < 3d
Choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 62
Consider an elementary reaction $$A(g)+B(g)\rightarrow C(g)+D(g)$$ If the volume of reaction mixture is suddenly reduced to $$\frac{1}{3}$$ of its initial volume, the reaction rate will become 'x' times of the original reaction rate. The value of x is :
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 63
The product formed in the following reaction sequence is :
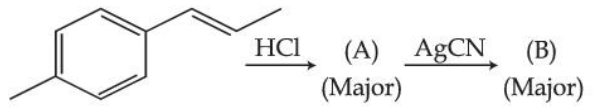
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 64
Assume a living cell with $$0.9%(\omega / \omega)$$ of glucose solution (aqueous). This cell is immersed in another solution having equal mole fraction of glucose and water. (Consider the data upto first decimal place only) The cell will
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 65
Given below are two statements : Statement (I): According to the Law of Octaves, the elements were arranged in the increasing order of their atomic number. Statement (II): Meyer observed a periodically repeated pattern upon plotting physical properties of certain elements against their respective atomic numbers. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 66
Match List - I with List - II.

Choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 67
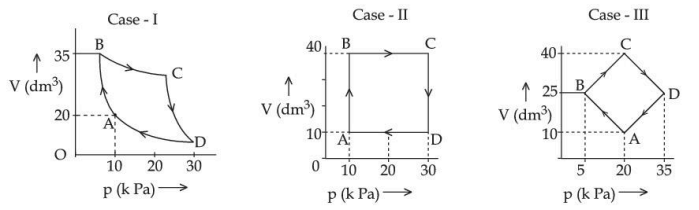
An ideal gas undergoes a cyclic transformation starting from the point A and coming back to the same point by tracing the path $$ A \rightarrow B \rightarrow C \rightarrow D \rightarrow A$$ as shown in the three cases above. Choose the correct option regarding $$\Delta U$$ :
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 68
Identify the inorganic sulphides that are yellow in colour : (A) $$\mathrm{(NH_4)_2S}$$ (B) $$\mathrm{PbS}$$ (C) $$\mathrm{CuS}$$ (D) $$\mathrm{As_2S_3}$$ (E) $$\mathrm{As_2S_5}$$ Choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 69
The major product of the following reaction is :
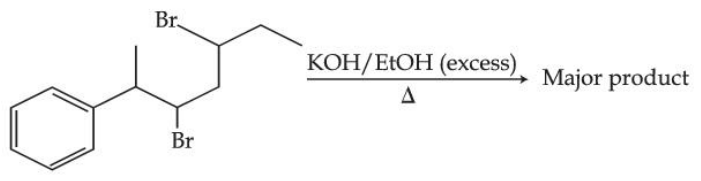
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 70
Identify correct conversion during acidic hydrolysis from the following : (A) starch gives galactose. (B) cane sugar gives equal amount of glucose and fructose. (C) milk sugar gives glucose and galactose. (D) amylopectin gives glucose and fructose. (E) amylose gives only glucose. Choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 71
Electrolysis of 600 mL aqueous solution of NaCl for 5 min changes the pH of the solution to 12 . The current in Amperes used for the given electrolysis is ____. (Nearest integer).
789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 72
A group 15 element forms $$d\pi-d\pi$$ bond with transition metals. It also forms hydride, which is a strongest base among the hydrides of other group members that form $$d\pi-d\pi$$ bond. The atomic number of the element is ____.
789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 73
Total number of molecules/species from following which will be paramagnetic is ______
$$O_{2}$$, $$O_2^+$$, $$O_2^-$$, $$NO$$, $$NO_{2}$$, $$CO$$, $$K_{2}[NiCl_{4}]$$, $$[Co(NH_{3})_{6}]Cl_{3}$$, $$K_{2}[Ni(CN)_{4}]$$
789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 74
Consider the following data : Heat of formation of $$CO_{2}$$(g)= -393.5 kJ $$mol^{-1}$$ Heat of formation of $$H_{2}0$$(l) = - 286.0 kJ $$mol^{-1}$$ Heat of combustion of benzene =-3267.0 kJ $$mol^{-1}$$ The heat of formation of benzene is _ . (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2025 Shift 2 - Chemistry - Question 75
The spin only magnetic moment ( $$\mu$$) value (B.M.) of the compound with strongest oxidising power among $$Mn_{2}O_{3},$$ TiO and VO is ____ B.M. (Nearest integer).
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




