NTA JEE Mains 28th Jan 2025 Shift 1
For the following questions answer them individually
NTA JEE Mains 28th Jan 2025 Shift 1 - Question 71
The formation enthalpies, $$\Delta H_{f}\ominus$$ for $$H_{g}$$ and $$O_{g}$$ are 220.0 and $$250.0 kJ mol^{-1}$$, respectively, at 298.15 K , and $$\Delta H_{f}\ominus$$ for $$H_{2}O_{g}$$ is $$-242.5 kJ mol^{-1}$$ at the same temperature. The average bond enthalpy of the $$O-H$$ bond in water at 298.15 K is __________$$kJ mol^{-1}$$(nearest integer).
789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2025 Shift 1 - Question 72
The molarity of a 70% (mass /mass) aqueous solution of a monobasic acid (X) is __________ $$\times10^{-1}$$ M(Nearest integer) [Given: Density of aqueous solution of (X) is 1.25 g $$mL^{-1}$$ Molar mass of the acid is $$70 g mol^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2025 Shift 1 - Question 73
Consider the following sequence of reactions:
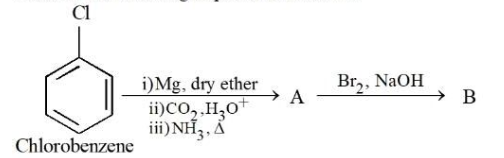
11.25 mg of chlorobenzene will produce __________$$\times 10^{-1}mg$$ of product B . (Consider the reactions result in complete conversion.) [Given molar mass of C, H, O, N and Cl as 12, 1, 16, 14 and $$35.5 g mol^{-1}$$ respectively]
789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2025 Shift 1 - Question 74
Quantitative analysis of an organic compound (X) shows following \% omposition. C : 14.5% Cl: 64.46\% H: 1.8 \%(Empirical formula mass of the compound (X) is__________$$\times 10^{-1}$$ (Given molar mass in $$gmil^{-1}$$ of C : 12, H : 1, O : 16, Cl : 35.5 )
789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2025 Shift 1 - Question 75
Given below is the plot of the molar conductivity vs $$\sqrt{concentration}$$ for KCl in aqueous solution.
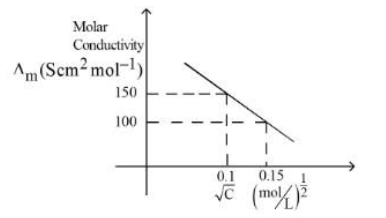
If, for the higher concentration of KCl solution, the resistance of the conductivity cell is 100Ω, then the resistance of the same cell with the dilute solution is $$'n'$$ Ω The value of $$x$$ is __________ (Nearest integer)
789
456
123
0.-
Clear All


