NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 51
Given below are two statements: Statement I : D-glucose pentaacetate reacts with 2, 4-dinitrophenylhydrazine Statement II : Starch, on heating with concentrated sulfuric acid at $$100^{\circ}$$ and 2-3 atmosphere pressure produces glucose. In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 52
Both acetaldehyde and acetone (individually) undergo which of the following reactions? A. Iodoform Reaction B. Cannizaro Reaction C. Aldol Condensation D. Tollen's Test E. Clemmensen Reduction Choose the correct answer from the options given below:
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 53
Which of the following oxidation reactions are carried out by both $$K_{2}Cr_{2}O_{7}$$ and $$KMnO_{4}$$ in acidic medium? $$A.\Gamma^{-}\rightarrow I_{2}B.S^{2-}\rightarrow S C.Fe^{2+}\rightarrow Fe^{3+}D.\Gamma^{-}\rightarrow IO_{3}^{-}E.S_{2}O_{3}^{2-}\rightarrow SO_{4}^{2-}$$ Choose the correct answer from the options given below:
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 54
A molecule (" P ") on treatment with acid undergoes rearrangement and gives ("Q"). ("Q") on ozonolysis followed by reflux under alkaline condition gives (" R "). The structure of (" R ") is given below.
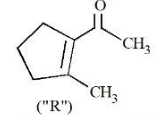
The structure of("P")is
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 55
The correct order of stability of following carbocations is :
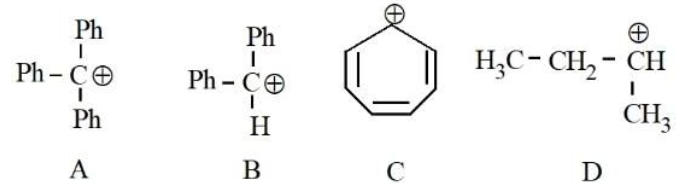
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 56
The products A and B in the following reactions, respectively are
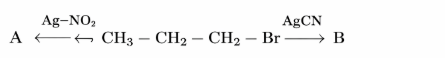
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 57
In a multielectron atom, which of the following orbitals described by three quantum numbers will have same energy in absence of electric and magnetic fields? $$A. n = 1, l = 0, m_{1}=0 B.n = 2,l = 0, m_{1}=0 C.n = 2,l = 1, m_{1}=1 D.n = 3,l = 2, m_{1} = 1 E.n = 3, l = 2, m_{1} = 0$$ Choose the correct answer from the options given below:
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 58
A weak acid HA has degree of dissociation x . Which option gives the correct expression of $$(pH pK_{a})$$ ?
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 59
The molecules having square pyramidal geometry are
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 60
Consider the following elements In, Tl, Al, Pb, Sn and Ge . The most stable oxidation states of elements with highest and lowest first ionisation enthalpies, respectively, are
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 61
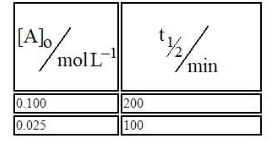
For a given reaction $$R\rightarrow P,t_{1/2}$$ is related to $$[A]_{\circ}$$ as given in table. Given: $$\log 2=0.30$$ Which of the following is true? A. The order of the reaction is 1/2.B.If $$[A]_{\circ}$$ is 1 M, then $$t_{1/2}$$ is $$200\sqrt{10}min$$ C.The order of the reaction changes to 1 if the concentration of reactant changes from 0.100 M to 0.500 M . D. $$t_{1/2}$$ is 800 min for $$[A]_{\circ}=1.6M$$ Choose the correct answer from the options given below: Options
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 62
Match the LIST-I with LIST-II
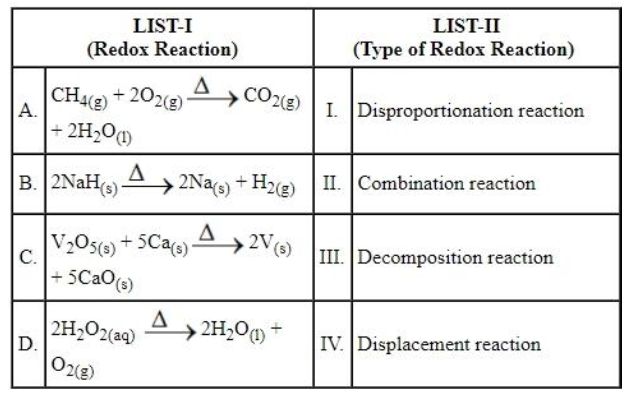
Choose the correct answer from the options given below:
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 63
Given below are two statements: Statement I: In the oxalic acid vs $$KMnO_{4}$$ (in the presence of dil $$H_{2}SO_{4}$$) titration the solution needs to be heated initially to $$60^{\circ}C$$, but no heating is required in Ferrous ammonium sulphate (FAS) vs $$KMnO_{4}$$ (in the presence of dil $$H_{2}SO_{4}$$) Statement II: In oxalic acid vs $$KMnO_{4}$$ titration, the initial formation of $$Mnso_{4}$$ takes place at high temperature, which then acts as catalyst for further reaction. In the case of FAS vs $$KMnO_{4}$$, heating oxidizes $$Fe^{2+}$$ and $$Fe^{3+}$$ by oxygen of air and error may be introduced in the experiment. In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 64
The metal ion whose electronic configuration is not affected by the nature of the ligand and which gives a violet colour in non-luminous flame under hot condition in borax bead test is
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 65
The compounds that produce $$CO_{2}$$ with aqueous $$NaHCO_{3}$$ solution are:
A.
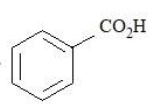
B.
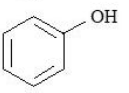
C.
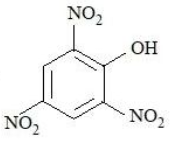
D.
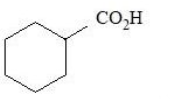
E.
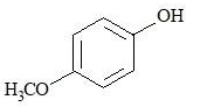
Choose the correct answer from the options given below:
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 66
What is the freezing point depression constant of a solvent, 50g of which contain 1 g non volatile solute (molar mass 256 g $$mol^{-1}$$) and the decrease in freezing point is 0.40 K ?
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 67
Given below are two statements:


involving lone pair of electrons on nitrogen. In the light of the above statements, choose the most appropriate answer from the options given below
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 68
Ice and water are placed in a closed container at a pressure of 1 atm and temperature 273.15 K . If pressure of the system is increased 2 times, keeping temperature constant, then identify correct observation from following
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 69
The incorrect decreasing order of atomic radii is
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 70
Consider $$'n'$$ is the number of lone pair of electrons present in the equatorial position of the most stable structure of $$ClF_{3}$$. The ions from the following with $$'n'$$ number of unpaired electrons are $$A. V^{3+} B.Ti^{3+} C.Cu^{2+} D.Ni^{2+} E.Ti^{2+}$$ Choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 71
The formation enthalpies, $$\Delta H_{f}\ominus$$ for $$H_{g}$$ and $$O_{g}$$ are 220.0 and $$250.0 kJ mol^{-1}$$, respectively, at 298.15 K , and $$\Delta H_{f}\ominus$$ for $$H_{2}O_{g}$$ is $$-242.5 kJ mol^{-1}$$ at the same temperature. The average bond enthalpy of the $$O-H$$ bond in water at 298.15 K is __________$$kJ mol^{-1}$$(nearest integer).
789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 72
The molarity of a 70% (mass /mass) aqueous solution of a monobasic acid (X) is __________ $$\times10^{-1}$$ M(Nearest integer) [Given: Density of aqueous solution of (X) is 1.25 g $$mL^{-1}$$ Molar mass of the acid is $$70 g mol^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 73
Consider the following sequence of reactions:
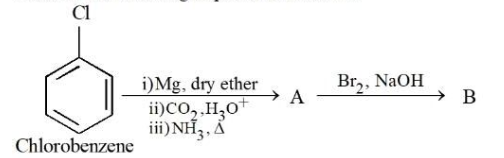
11.25 mg of chlorobenzene will produce __________$$\times 10^{-1}mg$$ of product B . (Consider the reactions result in complete conversion.) [Given molar mass of C, H, O, N and Cl as 12, 1, 16, 14 and $$35.5 g mol^{-1}$$ respectively]
789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 74
Quantitative analysis of an organic compound (X) shows following \% omposition. C : 14.5% Cl: 64.46\% H: 1.8 \%(Empirical formula mass of the compound (X) is__________$$\times 10^{-1}$$ (Given molar mass in $$gmil^{-1}$$ of C : 12, H : 1, O : 16, Cl : 35.5 )
789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2025 Shift 1 - Chemistry - Question 75
Given below is the plot of the molar conductivity vs $$\sqrt{concentration}$$ for KCl in aqueous solution.
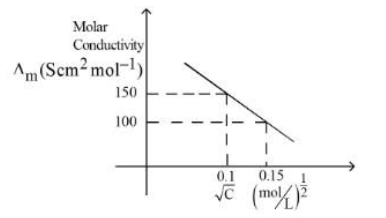
If, for the higher concentration of KCl solution, the resistance of the conductivity cell is 100Ω, then the resistance of the same cell with the dilute solution is $$'n'$$ Ω The value of $$x$$ is __________ (Nearest integer)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




