NTA JEE Mains 27th Jan 2024 Shift 1
For the following questions answer them individually
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 51
Mass of methane required to produce $$22$$ g of CO after complete combustion is _______ g. (Given Molar mass in g mol$$^{-1}$$, $$C = 12.0, H = 1.0, O = 16.0$$)
789
456
123
0.-
Clear All
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 52
The number of electrons present in all the completely filled subshells having $$n = 4$$ and $$s = +\frac{1}{2}$$ is ______ (Where $$n$$ = principal quantum number and $$s$$ = spin quantum number)
789
456
123
0.-
Clear All
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 53
Sum of bond order of CO and $$NO^+$$ is _______.
789
456
123
0.-
Clear All
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 54
If three moles of an ideal gas at $$300$$ K expand isothermally from $$30 \text{ dm}^3$$ to $$45 \text{ dm}^3$$ against a constant opposing pressure of $$80$$ kPa, then the amount of heat transferred is _________ J.
789
456
123
0.-
Clear All
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 55
Among the following, total number of meta directing functional groups is (Integer based) $$-OCH_3$$, $$-NO_2$$, $$-CN$$, $$-CH_3$$, $$-NHCOCH_3$$, $$-COR$$, $$-OH$$, $$-COOH$$, $$-Cl$$
789
456
123
0.-
Clear All
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 56
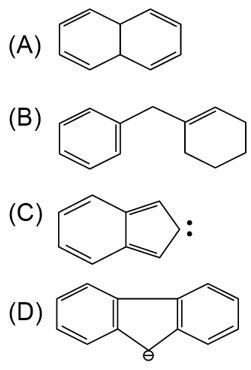
Among the given organic compounds, the total number of aromatic compounds is _______.
789
456
123
0.-
Clear All
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 57
3-Methylhex-2-ene on reaction with HBr in presence of peroxide forms an addition product (A). The number of possible stereoisomers for 'A' is _______.
789
456
123
0.-
Clear All
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 58
The mass of silver (Molar mass of Ag : $$108 \text{ g mol}^{-1}$$) displaced by a quantity of electricity which displaces $$5600$$ mL of $$O_2$$ at S.T.P. will be ______ g.
789
456
123
0.-
Clear All
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 59
Consider the following data for the given reaction $$2HI_{(g)} \rightarrow H_{2(g)} + I_{2(g)}$$. $$[HI] \text{ (mol L}^{-1}\text{)}$$: $$0.005, \; 0.01, \; 0.02$$. Rate $$\text{(mol L}^{-1} \text{ s}^{-1}\text{)}$$: $$7.5 \times 10^{-4}, \; 3.0 \times 10^{-3}, \; 1.2 \times 10^{-2}$$. The order of the reaction is _______.
789
456
123
0.-
Clear All
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 60
From the given list, the number of compounds with $$+4$$ oxidation state of Sulphur: $$SO_3, H_2SO_3, SOCl_2, SF_4, BaSO_4, H_2S_2O_7$$
789
456
123
0.-
Clear All


