NTA JEE Mains 27th Jan 2024 Shift 1
For the following questions answer them individually
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 31
The electronic configuration for Neodymium is: [Atomic Number for Neodymium 60]
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 32
Which of the following electronic configuration would be associated with the highest magnetic moment?
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 33
Choose the polar molecule from the following :
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 34
Which of the following is strongest Bronsted base?
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 35
Given below are two statements : Statement (I) : Aqueous solution of ammonium carbonate is basic.
Statement (II) : Acidic/basic nature of salt solution of a salt of weak acid and weak base depends on $$K_a$$ and $$K_b$$ value of acid and the base forming it.
In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 36
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Melting point of Boron (2453 K) is unusually high in group 13 elements.
Reason (R) : Solid Boron has very strong crystalline lattice.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 37
IUPAC name of the following compound (P) is :
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 38
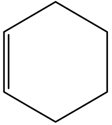
Cyclohexene is _________ type of an organic compound.
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 39
Which of the following has highly acidic hydrogen?
NTA JEE Mains 27th Jan 2024 Shift 1 - Question 40
The ascending order of acidity of $$-OH$$ group in the following compounds is :
(A) Bu-OH
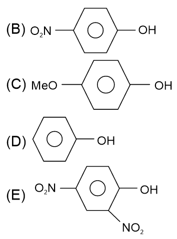
Choose the correct answer from the options given below :


