NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 51
The wavelength of spectral line obtained in the spectrum of $$Li^{2+}$$ ion, when the transition takes place between two levels whose sum is 4 and difference is 2, is
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 52
Find out the statements which are not true.
A. Resonating structures with more number of covalent bonds and lesser charge separntion are more stable.
B. In electromeric effect, an unsaturated system shows +E effect with nucleophile and -E effect with electrophile.
C. Inductive effect is responsible for high melting point, boiling point and dipole moment of polar compotmds.
D. The greater the number of alkyl groups attached to the doubly bonded carbon atoms, higher is the heat of hydrogenation.
E. Stability of carbanion increases with the increase in s - character of the carbon carrying the negative charge.
Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 53
"X" is an oxoanion of the lightest element of group 7 (in the periodic table). The metal is in +6 oxidation state in "X". The color of the potassium salt of X is
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 54
Consider the following gaseous equilibrimn in a closed container of volume 'V' at T(K).
$$P_{2}(g)+Q_{2}(g)\rightleftharpoons 2PQ(g)$$
2 moles each of $$P_{2}(g)$$, $$Q_{2}(g)$$ and PQ(g) are present at equilibrium. Now one mole each of'$$P_{2}$$' and '$$Q_{2}$$' are added to the equilibrium keeping the temperature at T(K). The number of moles of $$P_{2}$$, $$Q_{2}$$ and PQ at the new equilibrium, respectively, are
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 55
A student has planned to prepare acetanilide from aniline using acetic anhydride. The student has started from 9.3 g of aniline. However, the student has managed to obtain 11 g of dry acetanilide.
The % yield of this reaction is :-
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 56
From the following, how many compounds contain at least one secondary alcohol?
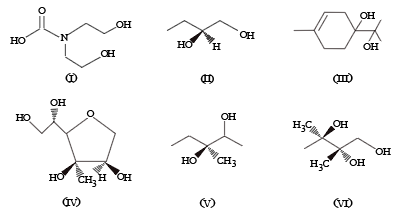
Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 57
Given below are two statements:
Statement I: The dipole moment of R-CN is greater than R-NC and R-NC can undergo hydrolysis under acidic medium to produce $$O\\||\\R-C-OH$$.
Statement II: R-CN hydrolyses under acidic medium to produce a compound which on treatment with $$SOCl_{2}$$, followed by the addition of $$NH_{3}$$ gives another compound(x). This compound (x) on treatment with NaOCl/NaOH gives a product, that on treatment with $$CHCl_{3}/KOH/ \Delta$$ produces R-NC
In the Light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 58
The heat of atomisation of methane and ethane are 'x' kJ $$mol^{-1}$$ and 'y' kJ $$mol^{-1}$$ respectively. The longest wavelength ($$\lambda$$) of light capable of breaking the C-C bond
can be expressed in SI unit as:
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 59
At 298 K, the mole percentage of $$N_{2}$$(g) in air is 80%. Water is in equilibrium with air at a pressure of 10 atm. What is the mole fraction of $$N_{2}$$(g) in water at 298 K?
($$K_{H}$$ for $$N_{2}$$ is $$6.5 \times 10^{7}$$ mm Hg)
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 60
Choose the INCORRECT statement
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 61
One mole of $$Cl_{2}(g)$$ was passed into 2 L of cold 2M KOH solution. After the reaction, the concentrations of $$Cl^{-}$$ , $$ClO^{-}$$ and $$OH^{-}$$ are respectively (assume volume remains constant)
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 62
Given below are two statements:
Statement I: Cross aldol condensation between two different aldehydes will always produce four different products.
Statement II: When semicarbazide reacts with a mixture of benzaldehyde and acetophenone under optimum pH, it fonns a condensation product with acetophenone only.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 63
Pair of species among the following having same bond order as well as paramagnetic character will be-
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 64
Given below are two statements:
Statement I: There are several conformers for n-butane. Out of those conformers,
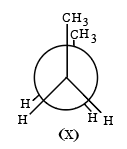
is the least stable and most stable conformer is
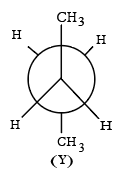
Statement II: As the dihedral angle increases, torsional strain decreases from (X) to (Y).
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 65
In the Group analysis of cations, $$Ba^{2+}$$ & $$Ca^{2+}$$ are precipitated respectively as
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 66
The number of possible tripeptides formed involving alanine (ala), glycine (gly) and valine (val), where no amino acid has been used more than once is:
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 67
Two liquids A and B form an ideal solution at temperature TK. At TK, the vapour pressures of pure A and B are 55 and 15 kN $$m^{-2}$$ respectively. What is the mole fraction of A in solution of A and B in equilibrium with a vapour in which the mole fraction of A is 0.8?
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 68
The wavelength of light absorbed for the following complexes are in the order
(I)$$\left[Co{(NH_{3})}_{6}\right]^{3+}$$
(II)$$\left[Co{(H_{2}O)}_{6}\right]^{3+}$$
(III)$$\left[Co{(CN)}_{6}\right]^{3-}$$
(IV)$$\left[Co(NH_{3})_{5}(H_{2}O)\right]^{3+}$$
(V)$$\left[CoF_{6}\right]^{3-}$$
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 69
The unsaturated ether On acidic hydrolysis produces carbonyl compom1ds as shown below:-
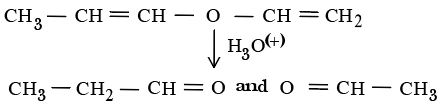
Based On this, predict the solution/reagent that will help to distillguish "P" and "Q" obtained in the following reaction:-
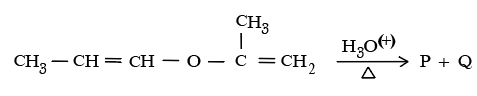
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 70
The correct order of C, N, 0 and F in terms of second ionisation potential is
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 71
0.25 g of an organic compound "A" containing carbon, hydrogen and oxygen was analysed using the combustion method. There was an increase in mass of $$CaCl_{2}$$ tube and potash tube at the end of the experiment. The amount was found to be 0.15 g and 0.1837 g, respectively. The percentage of oxygen in compound A is __ %. (Nearest integer)
(Given: molar massing $$mol^{-1}$$ H : 1, C : 12, O : 16)
789
456
123
0.-
Clear All
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 72
Molar conductivity of a weak acid HQ of concentration 0.18 M was found to be 1/30 of the molar conductivity of another weak acid HZ with concentration of 0.02M. If $$\lambda^{\circ}{}_{Q}-$$ happened to be equal with $$\lambda^{\circ}{}_{Z}-$$, then the difference of the $$pK_{a}$$ values of the two weak acids $$(pK_{a}(HQ) - pK_{a}(HZ))$$ is ___ (Nearest integer).
[Given: degree of dissociation ($$\alpha$$) << 1 for both weak acids, $$\lambda^{\circ}$$ : limiting molar conductivity of ions]
789
456
123
0.-
Clear All
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 73
A chromium complex with a formula $$CrCl_{3}.6H_{2}O$$ has a spin only magnetic moment value of 3.87 BM and its solution conductivity corresponds to 1:2 electrolyte. 2.75 g of the complex solution was initially passed through a cation exchanger. The solution obtained after the process was reacted with excess of $$AgNO_{3}$$. The amount of AgCI formed in the above process is __ g. (Nearest
integer)
[Given: Molar massing $$mol^{-1}$$ Cr:52; Cl:35.5, Ag:108, 0:16, H:1]
789
456
123
0.-
Clear All
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 74
Grignard reagent RMgBr (P) reacts with water and forms a gas (Q). One gram of Q occupies $$1.4 dm^{3}$$ at STP.(P) on reaction with dry ice in dry ether followed by $$H_{3}O^{+}$$ fonns a compound (Z). 0.1 mole of(Z) will weigh ____ g. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 24th Jan 2026 Shift 2 - Chemistry - Question 75
The half-life of $${}^{65}Zn$$ is 245 days. After x days, 75% of original activity remained. The value of x in days is ___ . (Nearest integer)
(Given: log 3 = 0.4771 and log 2 = 0.3010)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




