NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 51
For hydrogen atom, the orbital/s with lowest energy is/are:$$\text{ (A) } 4s \text{ (B) } 3p_x \text{ (C) } 3d_{x^2-y^2} \text{ (D) } 3d_{z^2} \text{ (E) } 4p_z$$ Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 52
Match List - I with List - II.
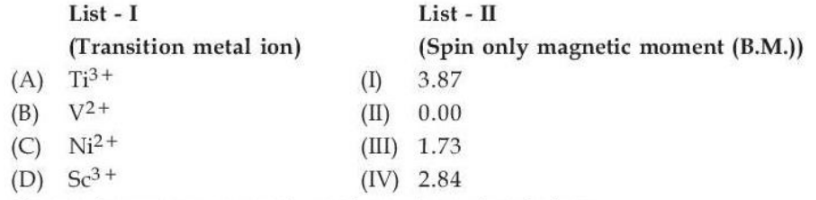
Choose the correct answer from the options given below :
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 53
Given below are two statements : Statement (I): Experimentally determined oxygen-oxygen bond lengths in the $$ O_3 $$ are found to be same and the bond length is greater than that of a $$O = O$$ (double bond) but less than that of a single $$(O - O)$$ bond. Statement (II) : The strong lone pair-lone pair repulsion between oxygen atoms is solely responsible for the fact that the bond length in ozone is smaller than that of a double bond $$(O = O)$$ but more than that of a single bond $$(O - O)$$. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 54
When Ethane-1,2-diamine is added progressively to an aqueous solution of Nickel (II) chloride, the sequence of colour change observed will be :
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 55
Given below are two statements :
Statement (I) : The first ionization energy of Pb is greater than that of Sn .
Statement (II) : The first ionization energy of Ge is greater than that of Si .
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 56
$$\text{Identify correct statement/s : }(A) -OCH_3 \text{ and } -NHCOCH_3 \text{ are activating groups. } (B)-CN \text{ and } -OH \text{ are meta directing groups. } (C) -CN \text{ and } -SO_3H$$ are meta directing groups. $$(D)$$ Activating groups act as ortho- and para-directing groups. $$(E)$$ Halides are activating groups. Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 57
Based on the data given below: $$\begin{aligned}E^\circ_{\text{Cr}_2\text{O}_7^{2-}/\text{Cr}^{3+}} &= 1.33 \text{V} &E^\circ_{\text{Cl}_2/\text{Cl}^-} &= 1.36\ \text{V} \\E^\circ_{\text{MnO}_4^-/\text{Mn}^{2+}} &= 1.51 \text{V} &E^\circ_{\text{Cr}^{3+}/\text{Cr}} &= -0.74\ \text{V}\end{aligned}$$ the strongest reducing agent is:
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 58
$$\text{For the reaction, } H_2(g) + I_2(g) \rightleftharpoons 2HI(g),$$ Attainment of equilibrium is predicted correctly by:
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 59
Find the compound ' A ' from the following reaction sequences.
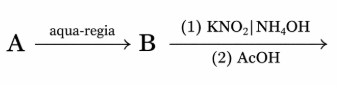
yellow ppt
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 60
The elemental composition of a compound is $$54.2\%C,\ 9.2\%H$$ and $$36.6\%O.$$ If the molar mass of the compound is $$132\ \text{g mol}^{-1},$$ the molecular formula of the compound is: [Given: Relative atomic masses C:H:O = 12:1:16]
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 61
{The conditions and consequence that favours the $$t_{2gg} e_g^1$$ configuration in a metal complex are:
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 62
In the given structure, the number of $$sp \text{ and } sp^2 $$ hybridized carbon atoms present respectively are:
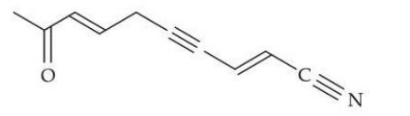
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 63
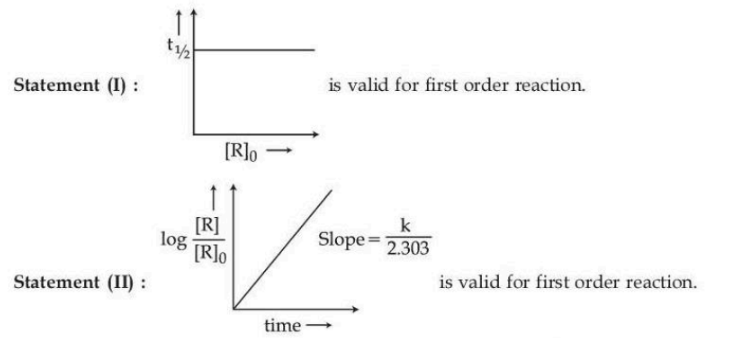
Given below are two statements :
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 64
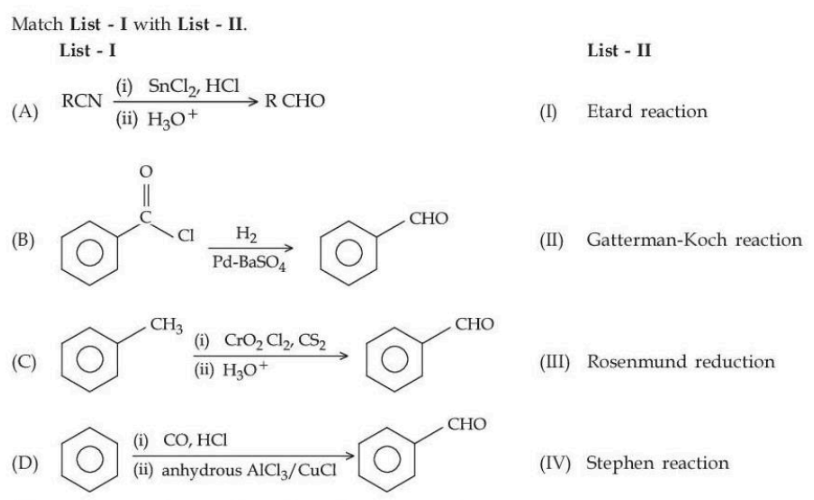
Choose the correct answer from the options given below :
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 65
The structure of the major product formed in the following reaction is :
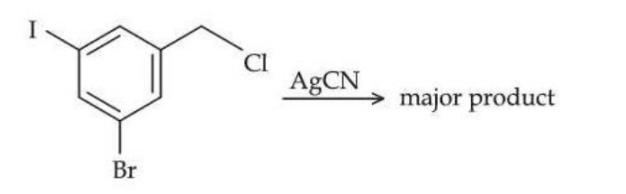
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 66
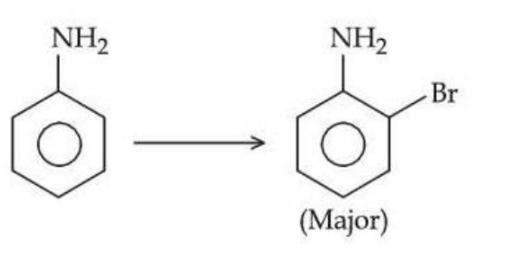
For reaction
The correct order of set of reagents for the above conversion is :
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 67
The successive 5 ionisation energies of an element are 800, 2427, 3658, 25024 and 32824 kJ/mol, respectively. By using the above values predict the group in which the above element is present:
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 68
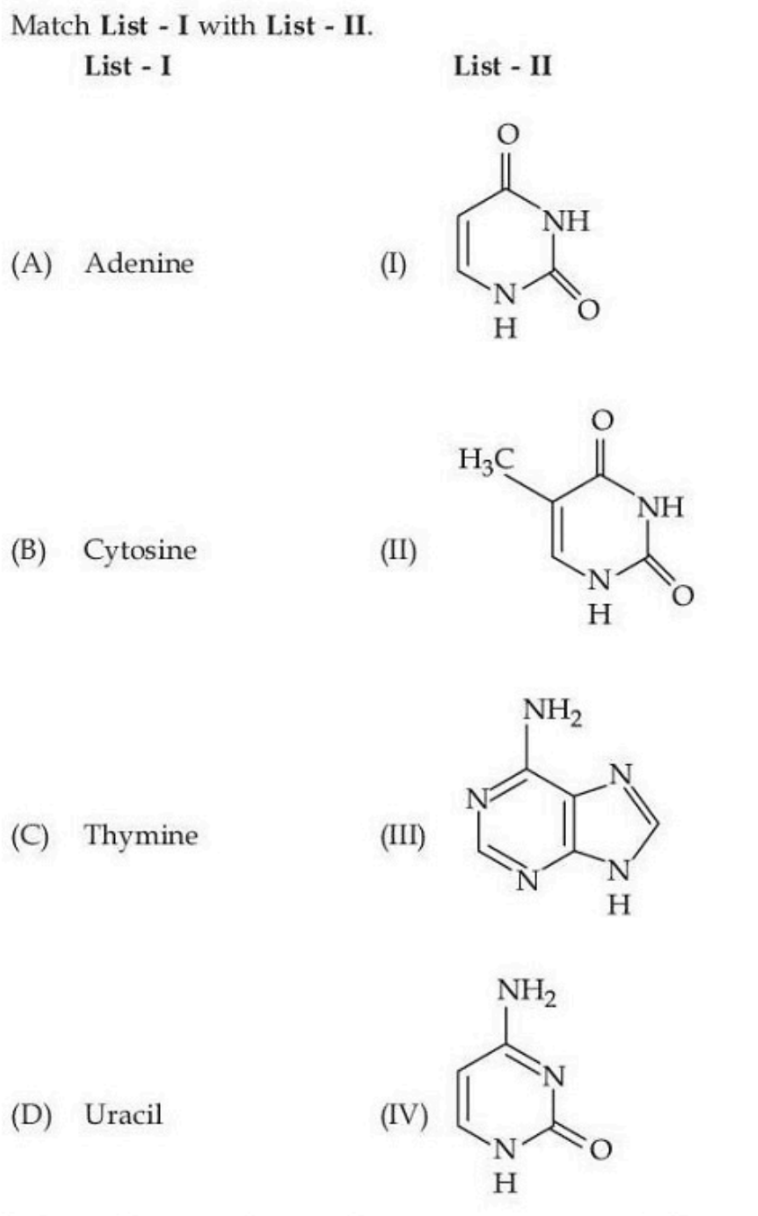
Choose the correct answer from the options given below :
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 69
Which of the following mixing of 1 M base and 1 M acid leads to the largest increase in temperature?
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 70
$$S(g) + \frac{3}{2}O_2(g) \rightarrow SO_3(g) + 2x\,\text{kcal} \\SO_2(g) + \frac{1}{2}O_2(g) \rightarrow SO_3(g) + y\,\text{kcal}\\\text{The heat of formation of } SO_2(g) \text{ is given by:}$$
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 71
In Carius method of estimation of halogen, $$0.25\,g$$ of an organic compound gave $$0.15\,g$$ of silver bromide $$(AgBr).$$ The percentage of Bromine in the organic compound is $$\underline{\hspace{2cm}}\times 10^{-1}$$ (Nearest integer). }[ Given: Molar mass of Ag is 108 and Br is 80 g $$mol^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 72
The observed and normal molar masses of compound $$MX_2$$ are $$65.6$$ and $$164$$ respectively. The percent degree of ionisation of $$MX_2$$ is $$\underline{\hspace{2cm}}\%.$$ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 73
Consider a complex reaction taking place in three steps with rate constants $$k_1,\; k_2 \text{ and } k_3$$ respectively. The overall rate constant $$k$$ is given by $$k=\sqrt{\frac{k_1k_3}{k_2}}$$. If the activation energies of the three steps are $$60,\;30$$ and $$10\,kJ\,mol^{-1}$$ respectively, then the overall energy of activation in $$kJ\,mol^{-1}$$ is $$\underline{\hspace{2cm}}.$$ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 74
The possible number of stereoisomers for 5-phenylpent -4-en-2-ol is $$\underline{\hspace{2cm}}.$$
789
456
123
0.-
Clear All
NTA JEE Mains 24th Jan 2025 Shift 2 - Chemistry - Question 75
The hydrocarbon $$(X)$$ with molar mass $$80\,g\,mol^{-1}$$ and $$90\% $$ carbon has $$\underline{\hspace{2cm}}$$ degree of unsaturation.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




