NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 51
The carbohydrate "Ribose" present in DNA, is A. A pentose sugar B. present in pyranose from C. in "D" configuration D. a reducing sugar, when free E. in $$\alpha$$-anomeric form Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 52
Given below are two statements: Statement I: The conversion proceeds well in the less polar medium.
$$\mathrm{CH_3-CH_2-CH_2-CH_2-Cl} \;\xrightarrow{\;\;HO^-\;\;} \mathrm{CH_3-CH_2-CH_2-CH_2-OH} + \mathrm{Cl^{(-)}}$$
Statement II: The conversion proceeds well in the more polar medium.
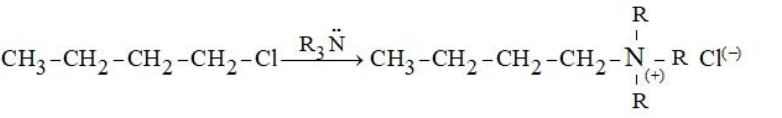
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 53
The product (A) formed in the following reaction sequence is
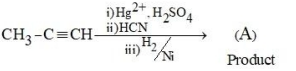
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 54
Aman has been asked to synthesise the molecule
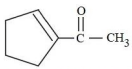
(x).He thought of preparing the molecule using an aldol condensation reaction. He found a few cyclic alkenes in his laboratory. He thought of performing ozonolysis reaction on alkene to produce a dicarbonyl compound followed by aldol reaction to prepare " x ". Predict the suitable alkene that can lead to the formation of " x ".
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 55
Which of the following arrangements with respect to their reactivity in nucleophilic addition reaction is correct?
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 56
Let us consider an endothermic reaction which is non-spontaneous at the freezing point of water. However, the reaction is spontaneous at boiling point of water. Choose the correct option.
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 57
Preparation of potassium permanganate from $$MnO_2$$ involves two step process in which the 1st step is a reaction with $$KOH$$ and $$KNO_3$$ to produce:
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 58
For a reaction, $$N_2O_5(g) \rightarrow 2NO_2(g) + \frac{1}{2}O_2(g)$$ in a constant volume container, no products were present initially. The final pressure of the system when $$50\%$$ of reaction gets completed is:
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 59
One mole of the octahedral complex compound $$Co(NH_3)_5Cl_3$$ gives 3 moles of ions on dissolution in water. One mole of the same complex reacts with excess $$AgNO_3$$ solution to yield two moles of $$AgCl(s).$$ The structure of the complex is:
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 60
Which of the following ions is the strongest oxidizing agent? (Atomic Number of $$Ce = 58,\; Eu = 63,\; Tb = 65,\; Lu = 71$$)
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 61
$$ K_{sp} \text{ for } Cr(OH)_3 \text{ is } 1.6\times10^{-30}.$$ What is the molar solubility of this salt in water?
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 62
Which of the following statements are NOT true about the periodic table? A. The properties of elements are function of atomic weights. B. The properties of elements are function of atomic numbers. C. Elements having similar outer electronic configurations are arranged in same period. D. An element's location reflects the quantum numbers of the last filled orbital. E. The number of elements in a period is same as the number of atomic orbitals available in energy level that is being filled. Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 63
Given below are two statements I and II. Statement I: Dumas method is used for estimation of "Nitrogen" in an organic compound. Statement II: Dumas method involves the formation of ammonium sulphate by heating the organic compound with conc $$H_2 SO_4 $$. In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 64
Which of the following statement is true with respect to $$H_2O,\ NH_3$$ and $$CH_4?$$ $$A.$$ The central atoms of all the molecules are $$sp^3$$ hybridized. $$B.$$ The $$H-O-H,\ H-N-H$$ and $$H-C-H$$ angles in the above molecules are $$104.5^\circ,\ 107.5^\circ$$ and $$109.5^\circ$$ respectively. $$C.$$ The increasing order of dipole moment is $$CH_4 < NH_3 < H_2O.$$ $$D.$$ Both $$H_2O$$ and $$NH_3$$ are Lewis acids and $$CH_4$$ is a Lewis base. $$E.$$ A solution of $$NH_3$$ in $$H_2O$$ is basic. In this solution $$NH_3$$ and $$H_2O$$ act as Lowry-Bronsted acid and base respectively. Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 65
Which one of the carbocations from the following is most stable?
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 66
Following are the four molecules "P", "Q", "R" and "S". Which one among the four molecules will react with
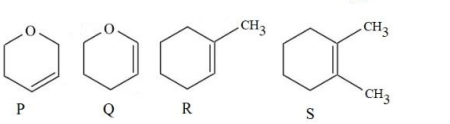
$$H-Br_{(aq)}$$ at the fastest rate?
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 67
For the given cell $$Fe^{2+}_{(aq)} + Ag^+_{(aq)} \rightarrow Fe^{3+}_{(aq)} + Ag_{(s)},$$ the standard cell potential of the above reaction is Given: $$\begin{aligned}Ag^+ + e^- &\rightarrow Ag \qquad E^\circ = x\,V \\Fe^{2+} + 2e^- &\rightarrow Fe \qquad E^\circ = y\,V \\Fe^{3+} + 3e^- &\rightarrow Fe \qquad E^\circ = z\,V \end{aligned}$$
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 68
The large difference between the melting and boiling points of oxygen and sulphur may be explained on the basis of
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 69
Consider the given plots of vapour pressure (VP) vs temperature (T/K). Which amongst the following options is correct graphical representation showing $$\Delta T_f,$$ depression in the freezing point of a solvent in a solution?
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 70
Which of the following linear combination of atomic orbitals will lead to formation of molecular orbitals in homonuclear diatomic molecules [internuclear axis in $$z$$-direction] ? A. $$2p_z$$ and $$2p_x$$ B. 2 s and $$2p_x$$ C. 3 $$d_{xy}$$ and 3 $$d_{x^{2} - y^{2}}$$ D. 2 s and $$2p_z$$ E. $$2p_z$$ and $$3d_{x}^{2}- y^{2}$$ Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 71
$$ X\,g $$ of benzoic acid on reaction with aq. $$NaHCO_3$$ released $$CO_2$$ that occupied $$11.2\,L$$ volume at STP. $$X$$ is $$\underline{\hspace{2cm}}\,g. $$
789
456
123
0.-
Clear All
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 72
Consider the following reaction occurring in the blast furnace: $$Fe_3O_4(s) + 4CO(g) \rightarrow 3Fe(l) + 4CO_2(g) x$$ kg of iron is produced when $$2.32\times10^3\,kg\,Fe_3O_4$$ and $$2.8\times10^2\,kg\,CO$$ are brought together in the furnace. The value of $$x$$ is $$\underline{\hspace{2cm}}$$ (nearest integer). Given: $$M(Fe_3O_4)=232\,g\,mol^{-1},$$ molar mass of $$CO=28\,g\,mol^{-1},$$ molar mass of $$(Fe)=56\,g\,mol^{-1}.$$
789
456
123
0.-
Clear All
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 73
$$ 37.8\,g\,N_2O_5$$ was taken in a $$1\,L$$ reaction vessel and allowed to undergo the following reaction at 500 K $$2N_2O_5(g) \rightleftharpoons 2N_2O_4(g) + O_2(g)$$ The total pressure at equilibrium was found to be $$18.65\,$$bar. Then, $$K_p = \underline{\hspace{2cm}}\times10^{-2}$$ [nearest integer]. Assume $$N_2O_5$$ to behave ideally under these conditions. Given: $$R=0.082\,bar\,L\,mol^{-1}K^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 74
Among the following cations, the number of cations which will give characteristic precipitate in their identification tests with $$K_4[Fe(CN)_6]$$ is $$\underline{\hspace{2cm}}$$. $$Cu^{2+},\; Fe^{3+},\; Ba^{2+},\; Ca^{2+},\; NH_4^{+},\; Mg^{2+},\; Zn^{2+}$$
789
456
123
0.-
Clear All
NTA JEE Mains 24th Jan 2025 Shift 1 - Chemistry - Question 75
Standard entropies of $$X_2,\ Y_2$$ and $$XY_5$$ are $$70,\ 50$$ and $$110\,J\,K^{-1}mol^{-1}$$ respectively. The temperature in Kelvin at which the reaction $$\frac{1}{2}X_2 + \frac{5}{2}Y_2 \rightleftharpoons XY_5 \Delta H^\ominus = -35\,kJ\,mol^{-1}$$ will be at equilibrium is $$\underline{\hspace{2cm}}$$ (Nearest integer).}
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




