NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 51
Identify the products [A] and [B], respectively in the following reaction :
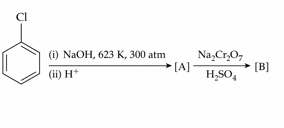
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 52
Consider the following reactions $$K_{2}Cr_{2}O_{7}\xrightarrow[-H_{2}O]{KOH}[A]\xrightarrow[-H_{2}O]{H_{2}SO_{4}}[B]+K_{2}SO_{4}$$ The products [A] and [B], respectively are :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 53
The effect of temperature on spontaneity of reactions are represented as :

NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 54
Which of the following graphs most appropriately represents a zero order reaction ?
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 55
Consider the reaction $$X_{2}Y(g)=X_{2}(g)+\frac{1}{2}Y_{2}(g)$$ The equation representing correct relationship between the degree of dissociation (x) of $$X_{2}Y(g)$$ with its equilibrium constant Kp is ______ . Assume x to be very very small.
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 56
Given below are two statements : Consider the following reaction
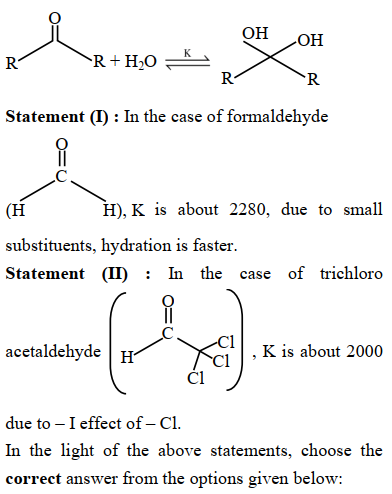
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 57
Given below are two statements : Statement (I) : For a given shell, the total number of allowed orbitals is given by $$n^{2}$$. Statement (II) : For any subshell, the spatial orientation of the orbitals is given by -l to +l values including zero. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 58
Standard electrode potentials for a few half cells are mentioned below :
$$E_{Cu^{2+}/Cu}^{\circ}=0.34 V,E_{Zn^{2+}/Zn}^{\circ}=-0.76 V\\E_{Ag^{+}/Ag}^{\circ}=0.80 V,E_{Mg^{2+}/Mg}^{\circ}=-2.37 V$$ Which one of the following cells gives the most negative value of $$\Delta G^{\circ}$$ ?
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 59
The $$\alpha$$-Helix and $$\beta$$- Pleated sheet structure of protein are associated with its :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 60
Given below are the atomic numbers of some group 14 elements. The atomic number of the element with lowest melting point is :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 61
Given below are two statements about X-ray spectra of elements : Statement (I) : A plot of $$\sqrt{v}$$($$\upsilon$$ = frequency of -rays emitted) vs atomic mass is a straight line. Statement (II) : A plot of $$\upsilon(\upsilon$$ = frequency of -rays emitted) vs atomic number is a straight line. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 62
Identify A,B and C in the given below reaction sequence
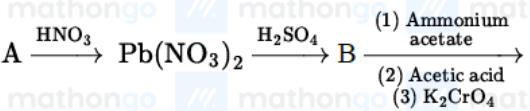
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 63
Given below are two statements : Statement (I) : The boiling points of alcohols and phenols increase with increase in the number of C-atoms. Statement (II) : The boiling points of alcohols and phenols are higher in comparison to other class of compounds such as ethers, haloalkanes. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 64
Consider a binary solution of two volatile liquid components 1 and 2.$$x_{1}$$ and $$y_{1}$$ are the mole fractions of component 1 in liquid and vapour phase, respectively. The slope and intercept of the linear plot of $$\frac{1}{x_{1}}$$ vs $$\frac{1}{y_{1}}$$ are given respectively as:
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 65
The ascending order of relative rate of solvolysis of following compounds is :
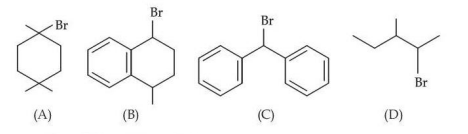
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 66
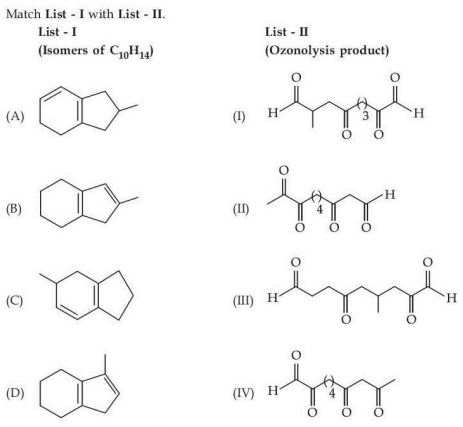
Match List - I with List - II.

Choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 67
Choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 68
pH of water is 7 at $$25^{\circ}C$$.If water is heated to $$80^{\circ}C$$.,it's pH will :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 69
Identify the coordination complexes in which the central metal ion has $$d^{4}$$ configuration.
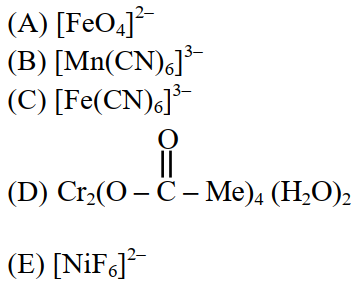
Choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 70
When a non-volatile solute is added to the solvent, the vapour pressure of the solvent decreases by 10 mm of Hg . The mole fraction of the solute in the solution is 0.2 . What would be the mole fraction of the solvent if decrease in vapour pressure is 20 mm of Hg ?
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 71
0.01 mole of an organic compound (X) containing 10% hydrogen, on complete combustion produced $$0.9_{g}H_{2}O$$. Molar mass of (X) is_____$$mol^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 72
A compound 'X' absorbs 2 moles of hydrogen and 'X' upon oxidation with $$KMnO_{4}|H^{+}$$ gives

The total number of $$\sigma$$ bonds present in the compound 'X' is ________.
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 73
When 81.0 g of aluminium is allowed to react with 128.0 g of oxygen gas, the mass of aluminium oxide produced in grams is_______ - (Nearest integer) Given : Molar mass of Al is 27.0 g $$mol^{-1}$$ Molar mass of O is 16.0 g $$mol^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 74
The bond dissociation enthalpy of $$X_2$$ (ΔHbond) calculated from the given data is ____________ $$kJmol^{-1}$$. (Nearest integer)
- $$M^+X^-$$(s) → $$M^+$$(g) +$$X^-$$(g) ΔH°lattice = 800 $$kJmol^{-1}$$
- M(s) → M(g) ΔH°sub = 100 $$kJmol^{-1}$$
- M(g) → $$M^+(g)+e^{_-}(g)$$ ΔHi = 500 $$kJmol^{-1}$$
- X(g) → $$e^-(g)→X^-(g)$$ ΔH°eg = -300 $$kJmol^{-1}$$
- M(g) + ½X2(g) → $$M^+X^-$$(s) ΔH°f = -400 $$kJmol^{-1}$$
[Given: ($$M^+X^-$$) is a pure ionic compound and X forms a diatomic molecule ($$X_2$$) in gaseous state]
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2025 Shift 2 - Chemistry - Question 75
Consider the following sequence of reactions.
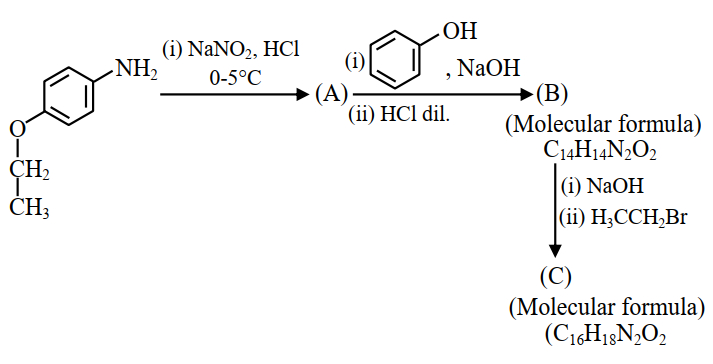
Total number of $$sp^{3}$$ hybridised carbon atoms in the major product C formed is________
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




