NTA JEE Mains 2nd April Shift 1 2026
For the following questions answer them individually
NTA JEE Mains 2nd April Shift 1 2026 - Question 61
Which of the following sets includes all the species that will change the orange colour of K$$_2$$Cr$$_2$$O$$_7$$ in acidic medium?
NTA JEE Mains 2nd April Shift 1 2026 - Question 62
Match List - I with List - II.

Choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 1 2026 - Question 63
Given below are two statements :
**Statement (I) :** 1,2,3-Trihydroxypropane can be separated from water by simple distillation.
**Statement (II) :** An azeotropic mixture cannot be separated by fractional distillation.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 1 2026 - Question 64
Given below are two statements :
**Statement (I) :** Benzyl chloride reacts faster in S$$_N$$1 mechanism than ethyl chloride.
**Statement (II) :** Ethyl carbocation intermediate is less stabilized by hyperconjugation than benzyl carbocation by resonance.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 1 2026 - Question 65
In IUPAC nomenclature, the correct order of decreasing priority of functional group is :
NTA JEE Mains 2nd April Shift 1 2026 - Question 66
For the given molecule, "x", the preferred site for the attack of the electrophile is :
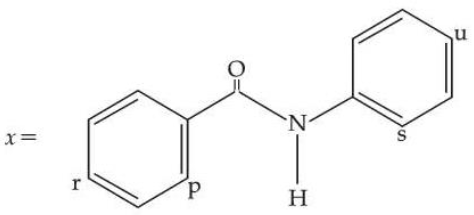
NTA JEE Mains 2nd April Shift 1 2026 - Question 67
Match List - I with List - II.

Choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 1 2026 - Question 68
Consider the three aromatic molecules (P, Q and R) whose structures have been given below :
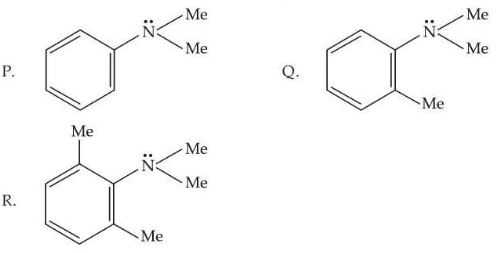
The correct order regarding the reactivity of these compounds with
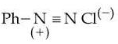
under optimum but slightly acidic medium is :
NTA JEE Mains 2nd April Shift 1 2026 - Question 69
Match List - I with List - II.

Choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 1 2026 - Question 70
A salt with few drops of conc. HCl gives apple green colour in flame test. The group precipitate of the salt is dissolved in acetic acid and treated with K$$_2$$CrO$$_4$$ to give yellow precipitate. When the sodium carbonate extract of the salt solution is heated with conc. HNO$$_3$$ and ammonium molybdate, it resulted a canary yellow precipitate. The cation and anion present in the salt are respectively,


