NTA JEE Mains 2nd April Shift 1 2026 - Chemistry
For the following questions answer them individually
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 51
The mass of iron converted into Fe$$_3$$O$$_4$$ by the action of 18 g of steam is : (Given : Molar mass of H, O and Fe are 1, 16 and 56 g mol$$^{-1}$$ respectively) Assume iron is present in excess :
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 52
What is the energy (in J atom$$^{-1}$$) required for the following process? $$\text{Li}^{2+}(g) \rightarrow \text{Li}^{3+}(g) + e^-$$ (Take the ionization energy for the H atom in the ground state as $$2.18 \times 10^{-18}$$ J atom$$^{-1}$$)
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 53
Given below are two statements : **Statement (I) :** The correct sequence of bond lengths in the following species is : $$O_2^+ < O_2 < O_2^- < O_2^{2-}$$ **Statement (II) :** The correct sequence of number of unpaired electrons in the following species is : $$O_2 > O_2^+ > O_2^- > O_2^{2-}$$ In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 54
Consider the following data.
(i) 2Al(s) + 6HCl(aq) → Al$$_2$$Cl$$_6$$(aq) + 3H$$_2$$(g) + 1200 kJ/mol
(ii) H$$_2$$(g) + Cl$$_2$$(g) → 2HCl(g) + 164 kJ/mol.
(iii) HCl(g) + aq → HCl(aq) + 83 kJ/mol.
(iv) Al$$_2$$Cl$$_6$$(s) + aq → Al$$_2$$Cl$$_6$$(aq) + 663 kJ/mol
The enthalpy of formation of anhydrous solid Al$$_2$$Cl$$_6$$ is :
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 55
19.5 g of fluoro acetic acid (molar mass = 78 g mol$$^{-1}$$) is dissolved in 500 g of water at 298 K. The depression in the freezing point was 1°C. What is K$$_a$$ of fluoro acetic acid? (For water, K$$_f$$ = 1.86 K kg mol$$^{-1}$$). Assume molarity and molality to have same values.
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 56
The solubility product constants of Ag$$_2$$CrO$$_4$$ and AgBr are 32x and 4y respectively at 298 K. The value of $$\left(\frac{\text{molarity of Ag}_2\text{CrO}_4}{\text{molarity of AgBr}}\right)$$ can be expressed as :
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 57
An electrochemical cell is constructed using half cells in the direction of spontaneous change
Fe(OH)$$_2$$(s) + 2e$$^-$$ → Fe(s) + 2OH$$^-$$(aq) E$$^0$$ = −0.88 V
and AgBr(s) + e$$^-$$ → Ag(s) + Br$$^-$$(aq) E$$^0$$ = +0.07 V
Which of the following option is correct?
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 58
$$t_{100\%}$$ is the time required for the 100% completion of the reaction while $$t_{1/2}$$ is the time required for 50% of the reaction to be completed. Which of the following option correctly represents the relation between $$t_{100\%}$$ and $$t_{1/2}$$ for zero and first order reactions respectively?
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 59
Given below are two statements : **Statement (I) :** The first ionisation enthalpy of the elements Na, Mg, Cl and Ar follows the order Na > Mg > Cl > Ar. **Statement (II) :** Among Ca, Al, Fe and B, the third ionisation enthalpy is very high for Ca. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 60
Given below are two statements : **Statement (I) :** Oxidising power of halogens decreases in the order F$$_2$$ > Cl$$_2$$ > Br$$_2$$ > I$$_2$$, which is the basis of "Layer test". **Statement (II) :** "Layer test" to identify Br$$_2$$ and I$$_2$$ in aqueous solution involves the oxidation of bromide or iodide into Br$$_2$$ or I$$_2$$ respectively with Cl$$_2$$, which is a type of displacement redox reaction. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 61
Which of the following sets includes all the species that will change the orange colour of K$$_2$$Cr$$_2$$O$$_7$$ in acidic medium?
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 62
Match List - I with List - II.

Choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 63
Given below are two statements :
**Statement (I) :** 1,2,3-Trihydroxypropane can be separated from water by simple distillation.
**Statement (II) :** An azeotropic mixture cannot be separated by fractional distillation.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 64
Given below are two statements :
**Statement (I) :** Benzyl chloride reacts faster in S$$_N$$1 mechanism than ethyl chloride.
**Statement (II) :** Ethyl carbocation intermediate is less stabilized by hyperconjugation than benzyl carbocation by resonance.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 65
In IUPAC nomenclature, the correct order of decreasing priority of functional group is :
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 66
For the given molecule, "x", the preferred site for the attack of the electrophile is :
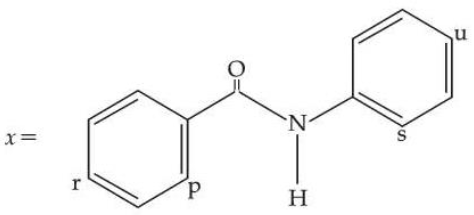
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 67
Match List - I with List - II.

Choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 68
Consider the three aromatic molecules (P, Q and R) whose structures have been given below :
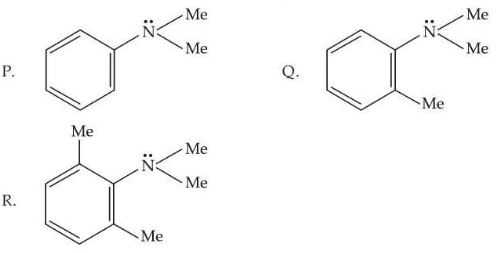
The correct order regarding the reactivity of these compounds with
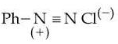
under optimum but slightly acidic medium is :
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 69
Match List - I with List - II.

Choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 70
A salt with few drops of conc. HCl gives apple green colour in flame test. The group precipitate of the salt is dissolved in acetic acid and treated with K$$_2$$CrO$$_4$$ to give yellow precipitate. When the sodium carbonate extract of the salt solution is heated with conc. HNO$$_3$$ and ammonium molybdate, it resulted a canary yellow precipitate. The cation and anion present in the salt are respectively,
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 71
5.33 g of CrCl$$_3$$·6H$$_2$$O, which is a 1 : 3 electrolyte, is dissolved in water and is passed through a cation exchanger. The chloride ions in the eluted solution, on treatment with AgNO$$_3$$ results in 8.61 g of AgCl. The ratio of moles of complex reacted and moles of AgCl formed is __________ × 10$$^{-2}$$. (Nearest integer) [Molar mass in g mol$$^{-1}$$ Cr : 52, Ag : 108, Cl : 35.5, H : 1, O : 16]
789
456
123
0.-
Clear All
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 72
Consider the isomers of hydrocarbon with molecular formula C$$_5$$H$$_{10}$$. These isomers do not decolourise KMnO$$_4$$ solution. These isomers are subjected to chlorination with chlorine in presence of light to give monochloro compounds. The total number of monochloro compounds (structural isomers only) formed is __________.
789
456
123
0.-
Clear All
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 73
One mole of an alkane (x) requires 8 mole oxygen for complete combustion. Sum of number of carbon and hydrogen atoms in the alkane (x) is __________.
789
456
123
0.-
Clear All
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 74
For reaction A → P, rate constant k = 1.5 × 10$$^3$$ s$$^{-1}$$ at 27°C. If activation energy for the above reaction is 60 kJ mol$$^{-1}$$, then the temperature (in °C) at which rate constant, k = 4.5 × 10$$^3$$ s$$^{-1}$$ is __________. (Nearest integer) Given : log 2 = 0.30, log 3 = 0.48, R = 8.3 J K$$^{-1}$$ mol$$^{-1}$$, ln 10 = 2.3
789
456
123
0.-
Clear All
NTA JEE Mains 2nd April Shift 1 2026 - Chemistry - Question 75
At the transition temperature T, A ⇌ B and ΔG$$^0$$ = 105 − 35 log T where A and B are two states of substance X. The transition temperature in °C when pressure is 1 atm is __________. (Nearest integer)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




