NTA JEE Mains 04th April 2024 Shift 1
For the following questions answer them individually
NTA JEE Mains 04th April 2024 Shift 1 - Question 31
Number of elements from the following that CANNOT form compounds with valencies which match with their respective group valencies is ______. B, C, N, S, O, F, P, Al, Si
NTA JEE Mains 04th April 2024 Shift 1 - Question 32
The correct order of first ionization enthalpy values of the following elements is: (A) O (B) N (C) Be (D) F (E) B. Choose the correct answer from the options given below :
NTA JEE Mains 04th April 2024 Shift 1 - Question 33
Which one of the following molecules has maximum dipole moment?
NTA JEE Mains 04th April 2024 Shift 1 - Question 34
Number of molecules/ions from the following in which the central atom is involved in sp³ hybridization is: $$NO_3^-,\ BCl_3,\ ClO_2^-,\ ClO_3$$
NTA JEE Mains 04th April 2024 Shift 1 - Question 35
Match List I with List II :
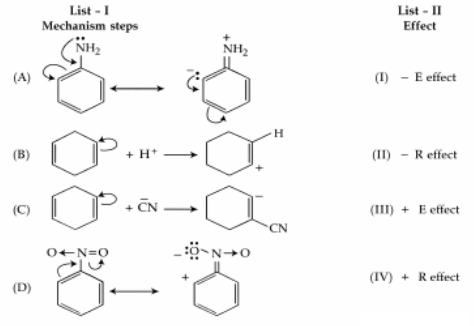
Choose the correct answer from the options given below :
NTA JEE Mains 04th April 2024 Shift 1 - Question 36
Which of the following nitrogen containing compound does not give Lassaigne's test?
NTA JEE Mains 04th April 2024 Shift 1 - Question 37
Which among the following is incorrect statement?
NTA JEE Mains 04th April 2024 Shift 1 - Question 38
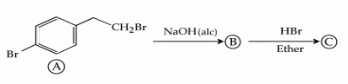
Identify (B) and (C) and how are (A) and (C) related ?
NTA JEE Mains 04th April 2024 Shift 1 - Question 39
The Molarity (M) of an aqueous solution containing 5.85 g of NaCl in 500 mL water is: (Given: Molar Mass Na: 23 and Cl: 35.5 g mol⁻¹)
NTA JEE Mains 04th April 2024 Shift 1 - Question 40
What pressure (bar) of H₂ would be required to make emf of hydrogen electrode zero in pure water at 25°C?


