NTA JEE Mains 04th April 2024 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 31
Number of elements from the following that CANNOT form compounds with valencies which match with their respective group valencies is ______. B, C, N, S, O, F, P, Al, Si
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 32
The correct order of first ionization enthalpy values of the following elements is: (A) O (B) N (C) Be (D) F (E) B. Choose the correct answer from the options given below :
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 33
Which one of the following molecules has maximum dipole moment?
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 34
Number of molecules/ions from the following in which the central atom is involved in sp³ hybridization is: $$NO_3^-,\ BCl_3,\ ClO_2^-,\ ClO_3$$
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 35
Match List I with List II :
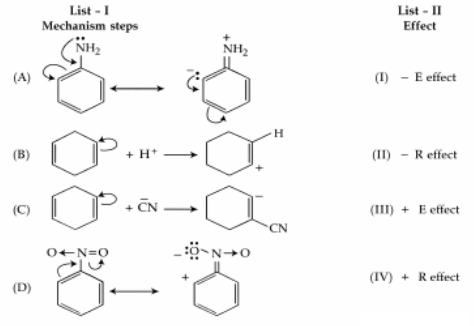
Choose the correct answer from the options given below :
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 36
Which of the following nitrogen containing compound does not give Lassaigne's test?
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 37
Which among the following is incorrect statement?
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 38
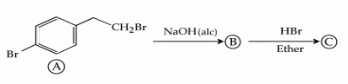
Identify (B) and (C) and how are (A) and (C) related ?
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 39
The Molarity (M) of an aqueous solution containing 5.85 g of NaCl in 500 mL water is: (Given: Molar Mass Na: 23 and Cl: 35.5 g mol⁻¹)
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 40
What pressure (bar) of H₂ would be required to make emf of hydrogen electrode zero in pure water at 25°C?
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 41
One of the commonly used electrode is calomel electrode. Under which of the following categories, calomel electrode comes?
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 42
What will be the decreasing order of basic strength of the following conjugate bases? $$^-OH,\ R\bar{O},\ CH_3CO\bar{O},\ C\bar{l}$$
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 43
The element which shows only one oxidation state other than its elemental form is:
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 44
Number of complexes from the following with even number of unpaired "d" electrons is: $$[V(H_2O)_6]^{3+}$$, $$\ [Cr(H_2O)_6]^{2+}$$, $$\ [Fe(H_2O)_6]^{3+}$$, $$\ [Ni(H_2O)_6]^{3+}$$, $$\ [Cu(H_2O)_6]^{2+}$$. [Given atomic numbers: V=23, Cr=24, Fe=26, Ni=28, Cu=29]
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 45
The correct sequence of ligands in the order of decreasing field strength is:
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 46
Identify the correct set of reagents or reaction conditions 'X' and 'Y' in the following set of transformation:
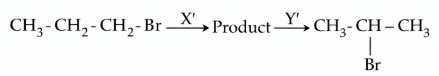
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 47
Given below are two statements: Statement I: Acidity of α-hydrogens of aldehydes and ketones is responsible for Aldol reaction. Statement II: Reaction between benzaldehyde and ethanal will NOT give Cross-Aldol product. Choose the most appropriate answer from the options given below :
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 48
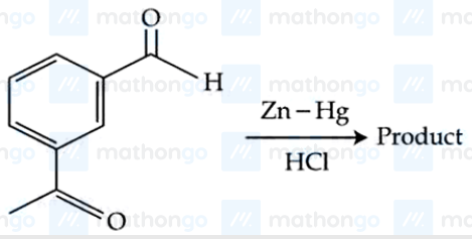
Identify the product in the following reaction :
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 49
In the precipitation of the iron group (III) in qualitative analysis, ammonium chloride is added before adding ammonium hydroxide to:
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 50
Which of the following is the correct structure of L-Glucose? (Four Fischer projection structures shown)
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 51
The de-Broglie's wavelength of an electron in the 4th orbit is ______ $$\pi a_0$$. ($$a_0$$ = Bohr's radius)
789
456
123
0.-
Clear All
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 52
Number of molecules/species from the following having one unpaired electron is ______. $$O_2,\ O_2^{-1},\ NO,\ CN^{-1},\ O_2^{2-}$$
789
456
123
0.-
Clear All
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 53
The enthalpy of formation of ethane (C₂H₆) from ethylene by addition of hydrogen where the bond-energies of C−H, C−C, C=C, H−H are 414 kJ, 347 kJ, 615 kJ and 435 kJ respectively is ______ kJ.
789
456
123
0.-
Clear All
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 54
Only 2 mL of KMnO₄ solution of unknown molarity is required to reach the end point of a titration of 20 mL of oxalic acid (2M) in acidic medium. The molarity of KMnO₄ solution should be ______ M.
789
456
123
0.-
Clear All
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 55
The number of different chain isomers for C₇H₁₆ is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 56
2.5 g of a non-volatile, non-electrolyte is dissolved in 100 g of water at 25°C. The solution showed a boiling point elevation by 2°C. Assuming the solute concentration is negligible with respect to the solvent concentration, the vapor pressure of the resulting aqueous solution is ______ mm of Hg (nearest integer). [Given: Kb = 0.52 K·kgmol⁻¹, 1 atm = 760 mm Hg, molar mass of water = 18 g mol⁻¹]
789
456
123
0.-
Clear All
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 57
Consider the following transformation involving first order elementary reaction in each step at constant
temperature as shown below.
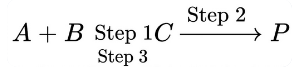
Some details of the above reactions are listed below.

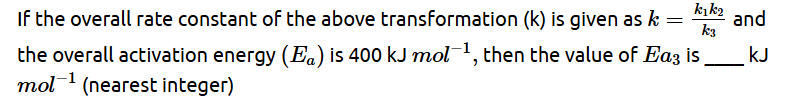
789
456
123
0.-
Clear All
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 58
Consider the following reaction: $$MnO_2 + KOH + O_2 \to A + H_2O$$. Product A in neutral or acidic medium disproportionates to give products B and C along with water. The sum of spin-only magnetic moment values of B and C is ______ BM. (nearest integer) [Given atomic number of Mn is 25]
789
456
123
0.-
Clear All
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 59
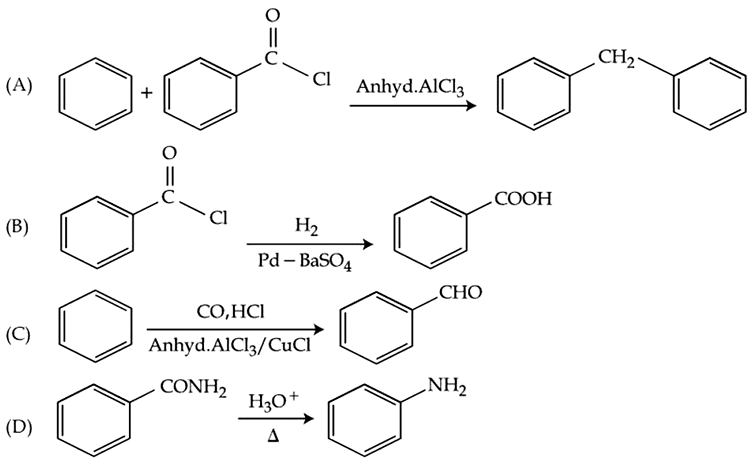
The number of the correct reaction(s) among the following is ______
789
456
123
0.-
Clear All
NTA JEE Mains 04th April 2024 Shift 1 - Chemistry - Question 60
X g of ethylamine is subjected to reaction with NaNO₂/HCl followed by water; evolved dinitrogen gas which occupied 2.24 L volume at STP. X is ______ × 10⁻¹ g.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




