NTA JEE Main 9th January 2020 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 26
The first and second ionisation enthalpies of a metal are 496 and 4560 kJ mol$$^{-1}$$, respectively. How many moles of HCl and H$$_2$$SO$$_4$$, respectively, will be needed to react completely with 1 mole of the metal hydroxide?
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 27
A mixture of gases $$O_2$$, $$H_2$$ and CO are taken in a closed vessel containing charcoal. The graph that represents the correct behaviour of pressure with time is:
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 28
The true statement amongst the following is:
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 29
In the figure shown below reactant A (represented by square) is in equilibrium with product B (represented by circle). The equilibrium constant is (approx):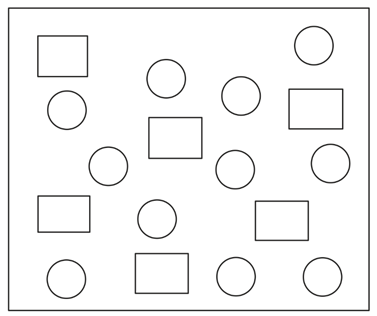
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 30
The solubility product of $$Cr(OH)_3$$ at 298K is $$6.0 \times 10^{-31}$$. The concentration of hydroxide ions in a saturated solution of $$Cr(OH)_3$$ will be:
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 31
5g of zinc is treated separately with an excess of
(a) dilute hydrochloric acid and
(b) aqueous sodium hydroxide.
The ratio of the volumes of $$H_2$$ evolved in these two reactions is:
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 32
Among the statements (a) - (d), the correct ones are:
(a) Lithium has the highest hydration enthalpy among the alkali metals.
(b) Lithium chloride is insoluble in pyridine.
(c) Lithium cannot form ethynide upon its reaction with ethyne.
(d) Both lithium and magnesium react slowly with $$H_2O$$.
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 33
The reaction of $$H_3N_3B_3Cl_3(A)$$ with $$LiBH_4$$ in tetrahydrofuran gives inorganic benzene (B). Further, the reaction of (A) with (C) leads to $$H_3N_3B_3(Me)_3$$. Compounds (B) and (C) respectively, are:
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 34
Which of the following has the shortest C - Cl bond?
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 35
Which of the following reactions will not produce a racemic product?
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 36
The number of $$sp^2$$ hybrid orbitals in a molecule of benzene is:
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 37
Biochemical Oxygen Demand (BOD) is the amount of oxygen required (in ppm):
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 38
Amongst the following, the form of water with the lowest ionic conductance at 298K is:
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 39
The correct order of the spin-only magnetic moments of the following complexes is:
(I) $$[Cr(H_2O)_6]Br_2$$
(II) $$Na_4[Fe(CN)_6]$$
(III) $$Na_3[Fe(C_2O_4)_3]$$ $$(\Delta_0 \gt P)$$
(IV) $$(Et_4N)_2[CoCl_4]$$
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 40
The isomer(s) of $$[Co(NH_3)_4Cl_2]$$ that has/have a Cl - Co - Cl angle of 90$$°$$, is/are:
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 41
In the following reaction A is: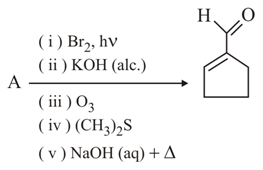
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 42
Consider the following reactions,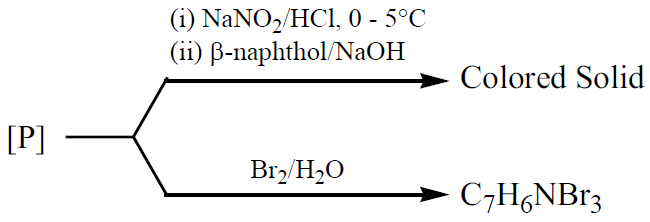
The compound [P] is:
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 43
The decreasing order of basicity of the following amines is: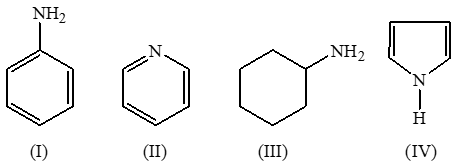
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 44
Which polymer has 'chiral' monomer(s)?
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 45
A, B and C are three biomolecules. The results of the tests performed on them are given below:
| Molisch's test | Barfoed Test | Biuret Test | |
|---|---|---|---|
| A | Positive | Negative | Negative |
| B | Positive | Positive | Negative |
| C | Negative | Negative | Positive |
A, B and C are respectively:
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 46
10.30 mg of $$O_2$$ is dissolved into a liter of sea water of density 1.03 g/mL. The concentration of $$O_2$$ in ppm is ___________.
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 47
A cylinder containing an ideal gas (0.1 mol of 1.0 dm$$^3$$) is in thermal equilibrium with a large volume of 0.5 molal aqueous solution of ethylene glycol at its freezing point. If the stoppers $$S_1$$ and $$S_2$$ (as shown in the figure) are suddenly withdrawn, the volume of the gas in litres after equilibrium is achieved will be ___________.
(Given, $$K_f$$(water) $$= 2.0$$ K kg mol$$^{-1}$$, R $$= 0.08$$ dm$$^3$$ atm K$$^{-1}$$ mol$$^{-1}$$)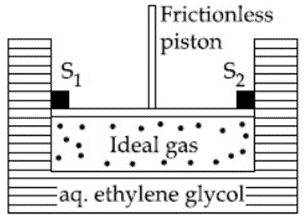
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 48
A sample of milk splits after 60 min at 300K and after 40 min at 400K when the population of lactobacillus acidophilus in it doubles. The activation energy (in kJ/mol) for this process is closest to ___________.
(Given, $$R = 8.3$$ J mol$$^{-1}$$ K$$^{-1}$$, $$\ln\left(\frac{2}{3}\right) = 0.4$$, $$e^{-3} = 4.0$$)
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 49
The sum of the total number of bonds between chromium and oxygen atoms in chromate and dichromate ions is ___________.
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 2 - Chemistry - Question 50
Consider the following reactions: $$A \xrightarrow[(ii)H_3O^+]{(i)CH_3MBr}B \xrightarrow[573K]{Cu} 2-methyl-2-butene$$. The mass percentage of carbon in A is ___________.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




