NTA JEE Main 9th January 2020 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 26
The de Broglie wavelength of an electron in the $$4^{th}$$ Bohr orbit is:
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 27
B has a smaller first ionization enthalpy than Be. Consider the following statement:
(I) it is easier to remove 2p electron than 2s electron
(II) 2p electron of B is more shielded from the nucleus by the inner core of electrons than the 2s electrons of Be
(III) 2s electron has more penetration power than 2p electron
(IV) atomic radius of B is more than Be
(atomic number B : 5, Be = 4)
The correct statements are:
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 28
The acidic, basic and amphoteric oxides, respectively, are:
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 29
If the magnetic moment of a di-oxygen species is 1.73 B.M., it may be:
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 30
If enthalpy of atomization for $$Br_2(l)$$ is $$x$$ kJ/mol and bond enthalpy for $$Br_2$$ is $$y$$ kJ/mol, the relation between them:
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 31
The $$K_{sp}$$ for the following dissociation is $$1.6 \times 10^{-5}$$
$$PbCl_{2(s)} \rightleftharpoons Pb^{2+}_{(aq)} + 2Cl^{-}_{(aq)}$$
Which of the following choices is correct for a mixture of 300 mL 0.134 M $$Pb(NO_3)_2$$ and 100 mL 0.4 M NaCl?
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 32
The compound that cannot act both as oxidizing and reducing agent is:
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 33
'X' melts at low temperature and is a bad conductor of electricity in both liquid and solid state. X is:
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 34
The increasing order of basicity for the following intermediates is (from weak to strong):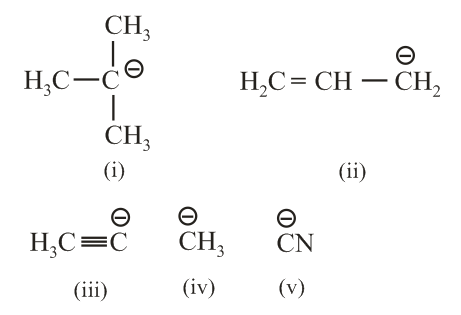
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 35
The correct order of heat of combustion for following alkadienes is: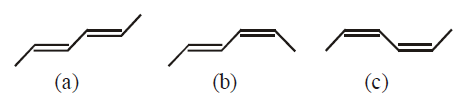
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 36
The major product (Y) in the following reactions is:
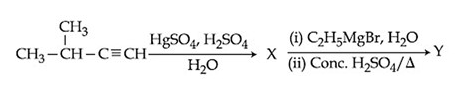
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 37
For the following reactions:
$$A \xrightarrow{700K} Product$$
$$A \xrightarrow[catalyst]{500K} Product$$
It was found that the $$E_a$$ is decreased by 30 KJ/mol in the presence of catalyst. If the rate remains unchanged, the activation energy for catalysed reaction is (Assume pre-exponential factor is same):
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 38
According to the following diagram, A reduces $$BO_2$$ when the temperature is: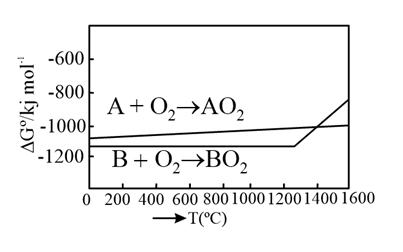
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 39
The electronic configurations of bivalent europium and trivalent cerium are:
(atomic number: Xe = 54, Ce = 58, Eu = 63)
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 40
Complex X of composition $$Cr(H_2O)_6Cl_n$$ has a spin only magnetic moment of 3.83 B.M. It reacts with $$AgNO_3$$ and shows geometrical isomerism. The IUPAC nomenclature of X is:
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 41
$$[Pd(F)(Cl)(Br)(I)]^{2-}$$ has $$n$$ number of geometrical isomers. Then, the spin-only magnetic moment and crystal field stabilization energy [CFSE] of $$[Fe(CN)_6]^{n-6}$$, respectively, are:
[Note: Ignore the pairing energy]
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 42
Which of these will produce the highest yield Friedel Crafts reaction?
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 43
Identify (A) in the following reaction sequence: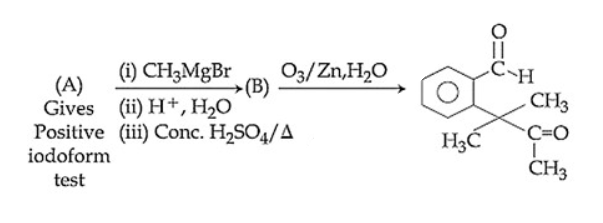
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 44
The major product Z obtained in the following reaction scheme is: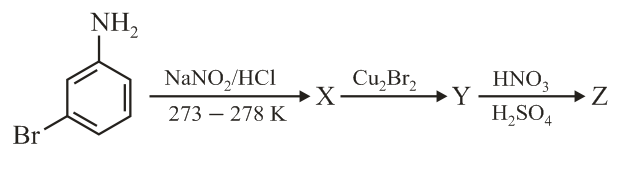
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 45
A chemist has 4 samples of artificial sweetener A, B, C and D. To identify these samples, he performed certain experiments and noted the following observations:
(i) A and D both form blue-violet colour with ninhydrin.
(ii) Lassaigne extract of C gives positive $$AgNO_3$$ test and negative $$Fe_4[Fe(CN)_6]_3$$ test.
(iii) Lassaigne extract of B and D gives positive sodium nitroprusside test.
Based on these observations which option is correct?
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 46
The molarity of $$HNO_3$$ in a sample which has density 1.4 g/mL and mass percentage of 63% is ___________.
(Molecular Weight of $$HNO_3 = 63$$)
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 47
The hardness of a water sample containing $$10^{-3}$$ M $$MgSO_4$$ expressed as $$CaCO_3$$ equivalents (in ppm) is ___________.
(molar mass of $$MgSO_4$$ is 120.37 g/mol)
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 48
How much amount of NaCl should be added to 600 g of water $$(\rho = 1.00 \; g/mL)$$ to decrease the freezing point of water to $$-0.2$$ °C? ___________. (The freezing point depression constant for water $$= 2$$ K $$kg \; mol^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 49
108 g of silver (molar mass 108 $$gmol^{-1}$$) is deposited at cathode from $$AgNO_3(aq)$$ solution by a certain quantity of electricity. The volume (in L) of oxygen gas produced at 273 K and 1 bar pressure from water by the same quantity of electricity is ___________.
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 1 - Chemistry - Question 50
The mass percentage of nitrogen in histamine is ___________.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




