NTA JEE Main 9th January 2019 Shift 2
For the following questions answer them individually
NTA JEE Main 9th January 2019 Shift 2 - Question 41
At 100$$^{\circ}$$C, copper (Cu) has FCC unit cell structure with cell edge length of $$x$$ $$\text{Å}$$. What is the approximate density of Cu (in g cm$$^{-3}$$) at this temperature? [Atomic Mass of Cu = 63.55 u]
NTA JEE Main 9th January 2019 Shift 2 - Question 42
A solution containing 62 g ethylene glycol in 250 g water is cooled to $$-10^{\circ}$$C. If $$K_f$$ for water is 1.86 K kg mol$$^{-1}$$, the amount of water (in g) separated as ice is:
NTA JEE Main 9th January 2019 Shift 2 - Question 43
If the standard electrode potential for a cell is 2 V at 300 K, the equilibrium constant (K) for the reaction.
$$Zn(s) + Cu^{2+}(aq) \rightleftharpoons Zn^{2+}(aq) + Cu(s)$$
at 300 K is approximately: ($$R = 8$$ J K$$^{-1}$$ mol$$^{-1}$$, $$F = 96000$$ C mol$$^{-1}$$)
NTA JEE Main 9th January 2019 Shift 2 - Question 44
For the reaction, $$2A + B \rightarrow$$ products, when the concentration of $$A$$ and $$B$$ both were doubled, the rate of the reaction increased from 0.3 mol L$$^{-1}$$ s$$^{-1}$$ to 2.4 mol L$$^{-1}$$ s$$^{-1}$$. When the concentration of $$A$$ alone is doubled, the rate increased from 0.3 mol L$$^{-1}$$ s$$^{-1}$$ to 0.6 mol L$$^{-1}$$ s$$^{-1}$$. Which one of the following statements is correct?
NTA JEE Main 9th January 2019 Shift 2 - Question 45
Consider the following reversible chemical reactions:
$$A_2(g) + B_2(g) \stackrel{k_1}{\rightleftharpoons} 2AB(g) \ldots (1)$$
$$6AB(g) \stackrel{k_2}{\rightleftharpoons} 3A_2(g) + 3B_2(g) \ldots (2)$$
The relation between $$K_1$$ and $$K_2$$ is:
NTA JEE Main 9th January 2019 Shift 2 - Question 46
For coagulation of arsenious sulphide sol, which of the following salt solutions will be most effective?
NTA JEE Main 9th January 2019 Shift 2 - Question 47
The correct match between Item I and Item II is:
Item I Item II
(A) Benzaldehyde (P) Mobile phase
(B) Alumina (Q) Adsorbent
(C) Acetonitrile (R) Adsorbate
NTA JEE Main 9th January 2019 Shift 2 - Question 48
The correct statement regarding the given Ellingham diagram is: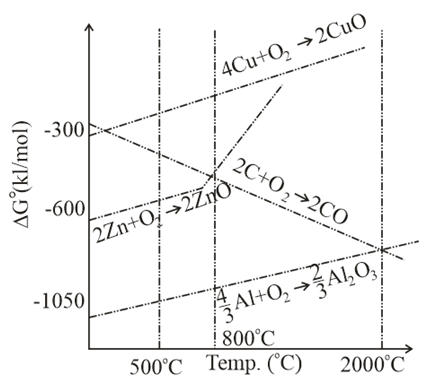
NTA JEE Main 9th January 2019 Shift 2 - Question 49
Good reducing nature of $$H_3PO_2$$ is attributed to the presence of:
NTA JEE Main 9th January 2019 Shift 2 - Question 50
The transition element that has the lowest enthalpy of atomisation is:


