NTA JEE Main 9th January 2019 Shift 1
For the following questions answer them individually
NTA JEE Main 9th January 2019 Shift 1 - Question 41
Arrange the following amines in the decreasing order of basicity.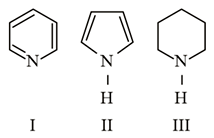
NTA JEE Main 9th January 2019 Shift 1 - Question 42
Which amongst the following is the strongest acid?
NTA JEE Main 9th January 2019 Shift 1 - Question 43
The compounds A and B in the following reaction are, respectively: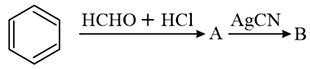
NTA JEE Main 9th January 2019 Shift 1 - Question 44
A water sample has ppm level concentration of the following metals: Fe = 0.2; Mn = 5.0; Cu = 3.0; Zn = 5.0. The metal that makes the water sample unsuitable for drinking is:
NTA JEE Main 9th January 2019 Shift 1 - Question 45
The one that is extensively used as a piezoelectric material is:
NTA JEE Main 9th January 2019 Shift 1 - Question 46
A solution of sodium sulphate contains 92 g of Na$$^+$$ ions per kilogram of water. The molality of Na$$^+$$ ions in that solution in mol kg$$^{-1}$$ is:
NTA JEE Main 9th January 2019 Shift 1 - Question 47
Which one of the following statements regarding Henry's law is not correct?
NTA JEE Main 9th January 2019 Shift 1 - Question 48
The anodic half-cell of lead-acid battery is recharged using electricity of 0.05 Faraday. The amount of $$PbSO_4$$ electrolyzed in g during the process is: (Molar mass of $$PbSO_4 = 303$$ g mol$$^{-1}$$)
NTA JEE Main 9th January 2019 Shift 1 - Question 49
The following results were obtained during kinetic studies of the reaction.
$$2A + B \rightarrow$$ product
Experiment I: A = 0.10 mol L$$^{-1}$$, B = 0.20 mol L$$^{-1}$$, Rate = $$6.93 \times 10^{-3}$$ mol L$$^{-1}$$ min$$^{-1}$$
Experiment II: A = 0.10 mol L$$^{-1}$$, B = 0.25 mol L$$^{-1}$$, Rate = $$6.93 \times 10^{-3}$$ mol L$$^{-1}$$ min$$^{-1}$$
Experiment III: A = 0.20 mol L$$^{-1}$$, B = 0.30 mol L$$^{-1}$$, Rate = $$1.386 \times 10^{-2}$$ mol L$$^{-1}$$ min$$^{-1}$$
The time (in minutes) required to consume half of A is:
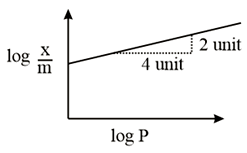
NTA JEE Main 9th January 2019 Shift 1 - Question 50
Adsorption of a gas follows Freundlich adsorption isotherm. In the given plot, $$x$$ is the mass of the gas adsorbed on mass $$m$$ of the adsorbent at pressure P. $$\frac{x}{m}$$ is proportional to: