NTA JEE Main 9th April 2016 Online
For the following questions answer them individually
NTA JEE Main 9th April 2016 Online - Question 41
Match the items in Column I with its main use listed in Column II:

NTA JEE Main 9th April 2016 Online - Question 42
The hydrocarbon with seven carbon atoms containing a neopentyl and a vinyl group is:
NTA JEE Main 9th April 2016 Online - Question 43
5 L of an alkane requires 25 L of oxygen for its complete combustion. If all volumes are measured at constant temperature and pressure, the alkane is
NTA JEE Main 9th April 2016 Online - Question 44
BOD stands for:
NTA JEE Main 9th April 2016 Online - Question 45
The solubility of $$N_2$$ in water at 300 K and 500 torr partial pressure is 0.01 g L$$^{-1}$$. The solubility (in g L$$^{-1}$$) at 750 torr partial pressure is:
NTA JEE Main 9th April 2016 Online - Question 46
What will happen when a block of copper metal is dropped into a beaker containing a solution of 1 M $$ZnSO_4$$?
NTA JEE Main 9th April 2016 Online - Question 47
The reaction of ozone with oxygen atoms in the presence of chlorine atoms can occur by a two step process shown below:
$$O_3(g) + Cl^\bullet \to O_2(g) + ClO^\bullet(g)$$ ...(i)
$$k_i = 5.2 \times 10^9$$ L mol$$^{-1}$$ s$$^{-1}$$
$$ClO^\bullet(g) + O^\bullet(g) \to O_2(g) + Cl^\bullet(g)$$ ...(ii)
$$k_{ii} = 2.6 \times 10^{10}$$ L mol$$^{-1}$$ s$$^{-1}$$
The closest rate constant for the overall reaction
$$O_3(g) + O^\bullet(g) \to 2O_2(g)$$ is:
NTA JEE Main 9th April 2016 Online - Question 48
A particular adsorption process has the following characteristics: (i) It arises due to van der Waals forces and (ii) it is reversible. Identify the correct statement that describes the above adsorption process:
NTA JEE Main 9th April 2016 Online - Question 49
The most appropriate method of making egg-albumin sol is:
NTA JEE Main 9th April 2016 Online - Question 50
The plot shows the variation of $$-\ln K_P$$ versus temperature for the two reactions.
$$M(s) + \frac{1}{2}O_2(g) \to MO(s)$$ and
$$C(s) + \frac{1}{2}O_2(g) \to CO(s)$$
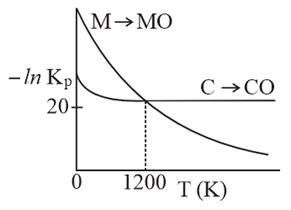
Identify the correct statement:


