NTA JEE Main 8th January 2020 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 26
Preparation of Bakelite proceeds via reactions:
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 27
The increasing order of the atomic radii of the following elements is:
(a) C
(b) O
(c) F
(d) Cl
(e) Br
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 28
Arrange the following bonds according to their average bond energies in descending order:
C-Cl, C-Br, C-F, C-I
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 29
Among the compounds A and B with molecular formula C$$_9$$H$$_{18}$$O$$_3$$, A is having higher boiling point than B. The possible structures of A and B are:
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 30
For the following Assertion and Reason, the correct option is:
Assertion: The pH of water increases with increase in temperature.
Reason: The dissociation of water into H$$^+$$ and OH$$^-$$ is an exothermic reaction.
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 31
Among the reactions (a) - (d), the reaction(s) that does/do not occur in the blast furnace during the extraction of iron is/are:
(a) CaO + SiO$$_2$$ $$\rightarrow$$ CaSiO$$_3$$
(b) 3Fe$$_2$$O$$_3$$ + CO $$\rightarrow$$ 2Fe$$_3$$O$$_4$$ + CO$$_2$$
(c) FeO + SiO$$_2$$ $$\rightarrow$$ FeSiO$$_3$$
(d) FeO $$\rightarrow$$ Fe + $$\frac{1}{2}$$O$$_2$$
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 32
The radius of the second Bohr orbit, in terms of the Bohr radius, $$a_0$$, in Li$$^{2+}$$ is:
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 33
Kjeldahl's method cannot be used to estimate nitrogen for which of the following compounds?
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 34
Consider the following plots of rate constant versus $$\frac{1}{T}$$ for four different reactions. Which of the following orders is correct for the activation energies of these reactions?
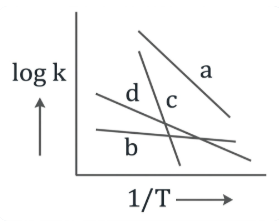
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 35
An unsaturated hydrocarbon X absorbs two hydrogen molecules on catalytic hydrogenation, and also gives following reaction:
X $$\xrightarrow[\frac{(ii) Zn}{H_2O}]{(i) O_3}$$ A $$\xrightarrow{[Ag(NH_3)_2]^+}$$ B (3-oxo-hexanedicarboxylic acid). X will be:
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 36
Which of the following compounds is likely to show both Frenkel and Schottky defects in its crystalline form?
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 37
Among (a) - (d), the complexes that can show geometrical isomerism are:
(a) [Pt(NH$$_3$$)$$_3$$Cl]$$^+$$
(b) [Pt(NH$$_3$$)Cl$$_5$$]$$^-$$
(c) [Pt(NH$$_3$$)$$_2$$Cl(NO$$_2$$)]
(d) [Pt(NH$$_3$$)$$_4$$ClBr]$$^{2+}$$
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 38
The major product in the following reaction is:
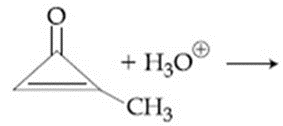
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 39
White phosphorus on reaction with concentrated NaOH solution in an inert atmosphere of CO$$_2$$ gives phosphine and compound (X). (X) on acidification with HCl gives compound (Y). The basicity of compound (Y) is:
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 40
Hydrogen has three isotopes (A), (B) and (C). If the number of neutron(s) in (A), (B) and (C) respectively, are (x), (y) and (z), the sum of (x), (y) and (z) is
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 41
A metal (A) on heating in nitrogen gas gives compound B. B on treatment with H$$_2$$O gives a colourless gas which when passed through CuSO$$_4$$ solution gives a dark blue-violet coloured solution. A and B respectively, are:
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 42
The major product [B] in the following sequence of reactions is:
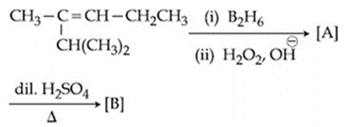
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 43
The correct order of the calculated spin-only magnetic moments of complexes (A) to (D) is:
(A) Ni(CO)$$_4$$
(B) [Ni(H$$_2$$O)$$_6$$]Cl$$_2$$
(C) Na$$_2$$[Ni(CN)$$_4$$]
(D) PdCl$$_2$$(PPh$$_3$$)$$_2$$
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 44
Two monomers in maltose are:
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 45
For the following Assertion and Reason, the correct option is:
Assertion: For hydrogenation reactions, the catalytic activity increases from Group 5 to Group 11 metals with maximum activity shown by Group 7-9 elements.
Reason: The reactants are most strongly adsorbed on group 7-9 elements.
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 46
NaClO$$_3$$ is used, even in spacecrafts, to produce O$$_2$$. The daily consumption of pure O$$_2$$ by a person in 492 L at 1 atm, 300K. How much amount of NaClO$$_3$$, in grams, is required to produce O$$_2$$ for the daily consumption of a person at 1 atm, 300K?
NaClO$$_3$$(s) + Fe(s) $$\rightarrow$$ O$$_2$$(g) + NaCl(s) + FeO(s)
R = 0.082 L atm mol$$^{-1}$$ K$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 47
At constant volume, 4 mol of an ideal gas when heated from 300K to 500K changes its internal energy by 5000J. The molar heat capacity at constant volume is
789
456
123
0.-
Clear All
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 48
In the following sequence of reactions the maximum number of atoms present in molecule 'C' in one plane is __________.
A $$\xrightarrow[Cu tube]{\text{Red hot }}$$ B $$\xrightarrow[\text{Anhydrous AlCl}_3]{CH_3Cl(1 eq)}$$ C
(A is a lowest molecular weight alkyne)
789
456
123
0.-
Clear All
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 49
For an electrochemical cell Sn(s)|Sn$$^{2+}$$(aq, 1M)||Pb$$^{2+}$$(aq, 1M)|Pb(s) the ratio $$\frac{[Sn^{2+}]}{[Pb^{2+}]}$$ when this cell attains equilibrium is __________.
(Given: E$$^0_{Sn^{2+}|Sn} = -0.14$$ V, E$$^0_{Pb^{2+}|Pb} = -0.13$$ V, $$\frac{2.303RT}{F} = 0.06$$)
789
456
123
0.-
Clear All
NTA JEE Main 8th January 2020 Shift 2 - Chemistry - Question 50
Complexes (ML$$_5$$) of metals Ni and Fe have ideal square pyramidal and trigonal bipyramidal geometries, respectively. The sum of the 90$$^\circ$$, 120$$^\circ$$ and 180$$^\circ$$ L-M-L angles in the two complexes is
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




