NTA JEE Main 8th January 2020 Shift 1
For the following questions answer them individually
NTA JEE Main 8th January 2020 Shift 1 - Question 31
The strength of an aqueous NaOH solution is most accurately determined by titrating: (Note: consider that an appropriate indicator is used)
NTA JEE Main 8th January 2020 Shift 1 - Question 32
When gypsum is heated to 393K, it forms:
NTA JEE Main 8th January 2020 Shift 1 - Question 33
Arrange the following compounds in increasing order of C-OH bond length: methanol, phenol, p-ethoxyphenol
NTA JEE Main 8th January 2020 Shift 1 - Question 34
A flask contains a mixture of isohexane and 3-methylpentane. One of the liquids boils at 63$$^\circ$$C while the other boils at 60$$^\circ$$C. What is the best way to separate the two liquids and which one will be distilled out first?
NTA JEE Main 8th January 2020 Shift 1 - Question 35
Among the gases (a) - (e), the gases that cause greenhouse effect are:
(a) CO$$_2$$
(b) H$$_2$$O
(c) CFCs
(d) O$$_2$$
(e) O$$_3$$
NTA JEE Main 8th January 2020 Shift 1 - Question 36
A graph of vapour pressure and temperature for three different liquids X, Y and Z is shown below: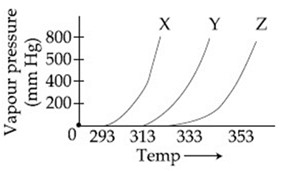
The following inferences are made:
(A) X has higher intermolecular interactions compared to Y.
(B) X has lower intermolecular interactions compared to Y.
(C) Z has lower intermolecular interactions compared to Y.
The correct inferences is/are:
NTA JEE Main 8th January 2020 Shift 1 - Question 37
The rate of a certain biochemical reaction at physiological temperature (T) occurs $$10^6$$ times faster with enzyme than without. The change in the activation energy upon adding enzyme is:
NTA JEE Main 8th January 2020 Shift 1 - Question 38
As per Hardy-Schulze formulation, the flocculation values of the following for ferric hydroxide sol are in the order:
NTA JEE Main 8th January 2020 Shift 1 - Question 39
The number of bonds between sulphur and oxygen atoms in S$$_2$$O$$_8^{2-}$$ and the number of bonds between sulphur and sulphur atoms in rhombic sulphur, respectively are:
NTA JEE Main 8th January 2020 Shift 1 - Question 40
The complex that can show fac- and mer-isomers is:


