NTA JEE Main 8th January 2020 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 26
For the Balmer series, in the spectrum of H atom, $$\bar{v} = R_H\left\{\frac{1}{n_1^2} - \frac{1}{n_2^2}\right\}$$, the correct statements among (I) to (IV) are,
(I) As wavelength decreases, the lines in the series converge.
(II) The integer $$n_1$$ is equal to 2.
(III) The lines of the longest wavelength correspond to $$n_2 = 3$$.
(IV) The ionization energy of hydrogen can be calculated from the wave number of these lines.
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 27
The first ionization energy (in kJ/mol) of Na, Mg, Al and Si respectively are:
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 28
The third ionization enthalpy is minimum for:
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 29
The predominant intermolecular forces present in ethyl acetate, a liquid, are:
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 30
The stoichiometry and solubility product of a salt with the solubility curve given below is, respectively: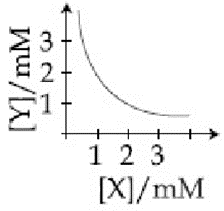
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 31
The strength of an aqueous NaOH solution is most accurately determined by titrating: (Note: consider that an appropriate indicator is used)
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 32
When gypsum is heated to 393K, it forms:
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 33
Arrange the following compounds in increasing order of C-OH bond length: methanol, phenol, p-ethoxyphenol
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 34
A flask contains a mixture of isohexane and 3-methylpentane. One of the liquids boils at 63$$^\circ$$C while the other boils at 60$$^\circ$$C. What is the best way to separate the two liquids and which one will be distilled out first?
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 35
Among the gases (a) - (e), the gases that cause greenhouse effect are:
(a) CO$$_2$$
(b) H$$_2$$O
(c) CFCs
(d) O$$_2$$
(e) O$$_3$$
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 36
A graph of vapour pressure and temperature for three different liquids X, Y and Z is shown below: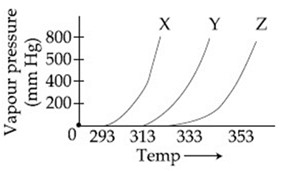
The following inferences are made:
(A) X has higher intermolecular interactions compared to Y.
(B) X has lower intermolecular interactions compared to Y.
(C) Z has lower intermolecular interactions compared to Y.
The correct inferences is/are:
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 37
The rate of a certain biochemical reaction at physiological temperature (T) occurs $$10^6$$ times faster with enzyme than without. The change in the activation energy upon adding enzyme is:
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 38
As per Hardy-Schulze formulation, the flocculation values of the following for ferric hydroxide sol are in the order:
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 39
The number of bonds between sulphur and oxygen atoms in S$$_2$$O$$_8^{2-}$$ and the number of bonds between sulphur and sulphur atoms in rhombic sulphur, respectively are:
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 40
The complex that can show fac- and mer-isomers is:
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 41
The decreasing order of reactivity towards dehydrohalogenation (E$$_1$$) reaction of the following compounds is:
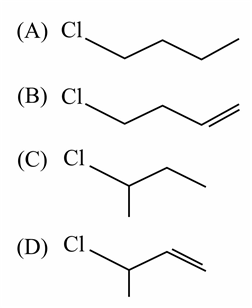
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 42
The major product of the following reaction is:
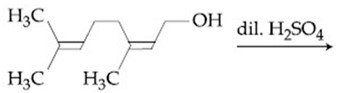
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 43
The most suitable reagent for the given conversion is:
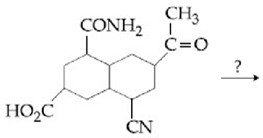
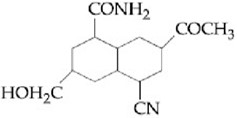
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 44
The major products A and B in the following reactions are
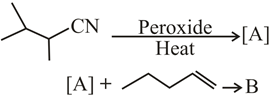
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 45
Which of the following statement is not true for glucose?
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 46
The volume (in mL) of 0.125 M AgNO$$_3$$ required to quantitatively precipitate chloride ions in 0.3 g of [Co(NH$$_3$$)$$_6$$]Cl$$_3$$ is __________.
M[Co(NH$$_3$$)$$_6$$]Cl$$_3$$ = 267.46 g/mol
M AgNO$$_3$$ = 169.87 g/mol
Report the nearest integer as the answer.
789
456
123
0.-
Clear All
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 47
The magnitude of work done by a gas that undergoes a reversible expansion along the path ABC shown in the figure is __________.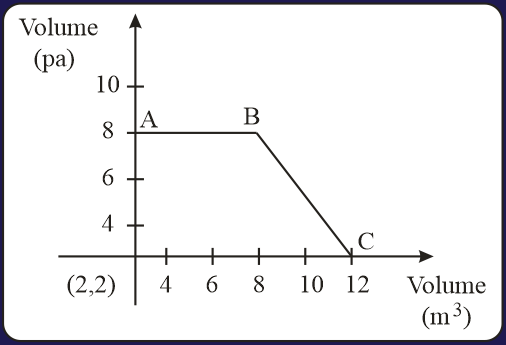
789
456
123
0.-
Clear All
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 48
Ferrous sulphate heptahydrate is used to fortify foods with iron. The amount (in grams) of the salt required to achieve 10 ppm of iron in 100 kg of wheat is __________.
Atomic weight: Fe = 55.85; S = 32.00; O = 16.00
789
456
123
0.-
Clear All
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 49
What would be the electrode potential for the given half-cell reaction at pH = 5?
2H$$_2$$O $$\rightarrow$$ O$$_2$$ + 4H$$^+$$ + 4e$$^-$$; E$$_{red}^0 = 1.23$$ V
(R = 8.314 J mol$$^{-1}$$ K$$^{-1}$$; Temp = 298K; oxygen under standard atm. pressure of 1 bar)
789
456
123
0.-
Clear All
NTA JEE Main 8th January 2020 Shift 1 - Chemistry - Question 50
The number of chiral centres in penicillin is __________.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




