NTA JEE Main 8th April2023 Shift 1
For the following questions answer them individually
NTA JEE Main 8th April2023 Shift 1 - Question 51
0.5 g of an organic compound (X) with 60% carbon will produce ______ $$\times 10^{-1}$$ g of CO$$_2$$ on complete combustion.
789
456
123
0.-
Clear All
NTA JEE Main 8th April2023 Shift 1 - Question 52
The number of following statement/s which is/are incorrect is ______
A) Line emission spectra are used to study the electronic structure
B) The emission spectra of atoms in the gas phase show a continuous spread of wavelength from red to violet.
C) An absorption spectrum is like the photographic negative of an emission spectrum
D) The element helium was discovered in the sun by spectroscopic method
789
456
123
0.-
Clear All
NTA JEE Main 8th April2023 Shift 1 - Question 53
The number of following factors which affect the percent covalent character of the ionic bond is______
A) Polarising power of cation
B) Extent of distortion of anion
C) Polarisability of the anion
D) Polarising power of anion
789
456
123
0.-
Clear All
NTA JEE Main 8th April2023 Shift 1 - Question 54
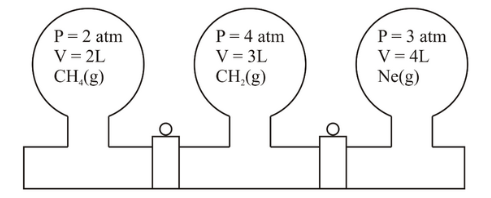
Three bulbs are filled with CH$$_4$$, CO$$_2$$ and Ne as shown in the picture. The bulbs are connected through pipes of zero volume. When the stopcocks are opened and the temperature is kept constant throughout, the pressure of the system is found to be ______ atm. (Nearest integer).
789
456
123
0.-
Clear All
NTA JEE Main 8th April2023 Shift 1 - Question 55
When a 60 W electric heater is immersed in a gas for 100 s in a constant volume container with adiabatic walls, the temperature of the gas rises by 5°C. The heat capacity of the given gas is JK$$^{-1}$$ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 8th April2023 Shift 1 - Question 56
The titration curve of weak acid vs. strong base with phenolphthalein as indicator is shown below. The $$K_{phenolphthalein} = 4 × 10^{−10}$$
Given: $$log 2 = 0.3$$
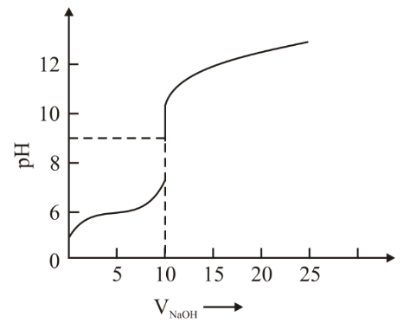
The number of following statement/s which is/are correct about phenolphthalein is ______
A. It can be used as an indicator for the titration of weak acid with weak base.
B. It begins to change colour at pH = 8.4
C. It is a weak organic base
D. It is colourless in acidic medium
789
456
123
0.-
Clear All
NTA JEE Main 8th April2023 Shift 1 - Question 57
Molar mass of the hydrocarbon (X) which on ozonolysis consumes one mole of O$$_3$$ per mole of (X) and gives one mole each of ethanal and propanone is ______ g mol$$^{-1}$$ (Molar mass of C: 12 g mol$$^{-1}$$, H: 1 g mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 8th April2023 Shift 1 - Question 58
The vapour pressure vs. temperature curve for a solution solvent system is shown below.
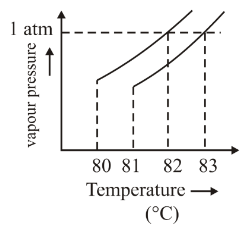
The boiling point of the solvent is ______°C.
789
456
123
0.-
Clear All
NTA JEE Main 8th April2023 Shift 1 - Question 59
The number of given statement/s which is/are correct is______
(A) The stronger the temperature dependence of the rate constant, the higher is the activation energy.
(B) If a reaction has zero activation energy, its rate is independent of temperature.
(C) The stronger the temperature dependence of the rate constant, the smaller is the activation energy.
(D) If there is no correlation between the temperature and the rate constant then it means that the reaction has negative activation energy.
789
456
123
0.-
Clear All
NTA JEE Main 8th April2023 Shift 1 - Question 60
XeF$$_4$$ reacts with SbF$$_5$$ to form [XeF$$_m$$]$$^{n+}$$[SbF$$_y$$]$$^{2-}$$. $$m + n + y + z$$ = ?
789
456
123
0.-
Clear All


