NTA JEE Main 8 April 2018 Offline
For the following questions answer them individually
NTA JEE Main 8 April 2018 Offline - Question 51
The compound that does not produce nitrogen gas by thermal decomposition is:
NTA JEE Main 8 April 2018 Offline - Question 52
Consider the following reaction and statements:
$$[Co(NH_3)_4Br_2]^+ + Br^- \rightarrow [Co(NH_3)_3Br_3] + NH_3$$
(i) Two isomers are produced if the reactant complex ion is a cis-isomer.
(ii) Two isomers are produced if the reactant complex ion is a trans-isomer.
(iii) Only one isomer is produced if the reactant complex ion is a trans-isomer.
(iv) Only one isomer is produced if the reactant complex ion is a cis-isomer.
The correct statements are:
NTA JEE Main 8 April 2018 Offline - Question 53
The oxidation states of Cr in [Cr(H$$_2$$O)$$_6$$]Cl$$_3$$, [Cr(C$$_6$$H$$_6$$)$$_2$$] and K$$_2$$[Cr(CN)$$_2$$(O)$$_2$$(O$$_2$$)(NH$$_3$$)], respectively, are:
NTA JEE Main 8 April 2018 Offline - Question 54
The major product of the following reaction is: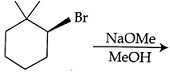
NTA JEE Main 8 April 2018 Offline - Question 55
Phenol reacts with methyl chloroformate in the presence of NaOH to form product A. A reacts with Br$$_2$$ to form product B. A and B are respectively:
NTA JEE Main 8 April 2018 Offline - Question 56
The major product formed in the following reaction is: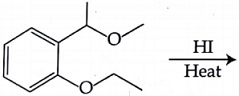
NTA JEE Main 8 April 2018 Offline - Question 57
Phenol on treatment with $$CO_2$$ in the presence of NaOH followed by acidification produces compound X as the major product. X on treatment with $$(CH_3CO)_2O$$ in the presence of catalytic amount of $$H_2SO_4$$ produces:
NTA JEE Main 8 April 2018 Offline - Question 58
The increasing order of basicity of the following compounds is: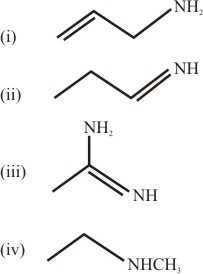
NTA JEE Main 8 April 2018 Offline - Question 59
Glucose on prolonged heating with HI gives:
NTA JEE Main 8 April 2018 Offline - Question 60
The predominant form of histamine present in human blood is (pK$$_a$$, Histidine = 6.0):


