NTA JEE Main 8 April 2018 Offline - Chemistry
For the following questions answer them individually
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 31
The ratio of mass percent of C and H of an organic compound ($$C_XH_YO_Z$$) is 6 : 1. If one molecule of the above compound ($$C_XH_YO_Z$$) contains half as much oxygen as required to burn one molecule of compound $$C_XH_Y$$ completely to $$CO_2$$ and $$H_2O$$. The empirical formula of the compound $$C_XH_YO_Z$$ is:
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 32
According to molecular orbital theory, which of the following molecule will not be available?
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 33
Which of the following compounds contain(s) no covalent bond(s)?
KCl, PH$$_3$$, O$$_2$$, B$$_2$$H$$_6$$, H$$_2$$SO$$_4$$
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 34
Total number of lone pair of electrons in I$$_3^-$$ ion is:
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 35
The combustion of benzene (l) gives CO$$_2$$(g) and H$$_2$$O(l). Given that heat of combustion of benzene at constant volume is $$-3263.9$$ kJ mol$$^{-1}$$ at 25$$^\circ$$C; the heat of combustion (in kJ mol$$^{-1}$$) of benzene at constant pressure will be:
(R = 8.314 JK$$^{-1}$$ mol$$^{-1}$$)
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 36
Which of the following lines correctly show the temperature dependence of equilibrium constant K, for an exothermic reaction?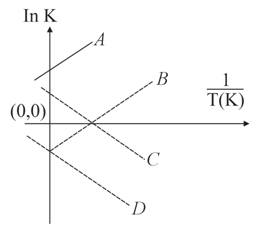
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 37
An aqueous solution contains 0.10 M H$$_2$$S and 0.20 M HCl. If the equilibrium constant for the formation of HS$$^-$$ from H$$_2$$S is $$1.0 \times 10^{-7}$$ and that of S$$^{2-}$$ from HS$$^-$$ ions is $$1.2 \times 10^{-13}$$, then the concentration of S$$^{2-}$$ ions in the aqueous solution is:
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 38
An aqueous solution contains an unknown concentration of Ba$$^{2+}$$. When 50 mL of a 1 M solution of Na$$_2$$SO$$_4$$ is added, BaSO$$_4$$ just begins to precipitate. The final volume is 500 mL. The solubility product of BaSO$$_4$$ is $$1 \times 10^{-10}$$. What is the original concentration of Ba$$^{2+}$$?
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 39
Which of the following are Lewis acids?
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 40
Which of the following salts is the most basic in aqueous solution?
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 41
An alkali is titrated against acid with methyl orange as an indicator, which of the following is a correct combination?
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 42
Hydrogen peroxide oxidises [Fe(CN)$$_6$$]$$^{4-}$$ to [Fe(CN)$$_6$$]$$^{3-}$$ in acidic medium, but reduces [Fe(CN)$$_6$$]$$^{3-}$$ to [Fe(CN)$$_6$$]$$^{4-}$$ in alkaline medium. The other products formed are, respectively:
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 43
When metal M is treated with NaOH, a white gelatinous precipitate X is obtained, which is soluble in excess of NaOH. Compound X when heated strongly gives an oxide which is used in chromatography as an adsorbent. The metal M is:
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 44
Which of the following compounds will be suitable for Kjeldahl's method for nitrogen estimation?
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 45
The trans-alkenes are formed by the reduction of alkynes with:
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 46
The recommended concentration of fluoride ion in drinking water is up to 1 ppm as fluoride ion is required to make teeth enamel harder by converting [3Ca$$_3$$(PO$$_4$$)$$_2$$.Ca(OH)$$_2$$] to:
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 47
Which type of 'defect' has the presence of cations in the interstitial sites?
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 48
For 1 molal aqueous solution of the following compounds, which one will show the highest freezing point?
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 49
How long (approximate) should water be electrolysed by passing through 100 amperes current so that the oxygen released can completely burn 27.66 g of diborane? (Atomic weight of B = 10.8 u)
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 50
At 518$$^\circ$$C, the rate of decomposition of a sample of gaseous acetaldehyde, initially at a pressure of 363 Torr was 1.00 Torr s$$^{-1}$$ when 5% had reacted and 0.50 Torr s$$^{-1}$$ when 33% had reacted. The order of the reaction is:
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 51
The compound that does not produce nitrogen gas by thermal decomposition is:
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 52
Consider the following reaction and statements:
$$[Co(NH_3)_4Br_2]^+ + Br^- \rightarrow [Co(NH_3)_3Br_3] + NH_3$$
(i) Two isomers are produced if the reactant complex ion is a cis-isomer.
(ii) Two isomers are produced if the reactant complex ion is a trans-isomer.
(iii) Only one isomer is produced if the reactant complex ion is a trans-isomer.
(iv) Only one isomer is produced if the reactant complex ion is a cis-isomer.
The correct statements are:
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 53
The oxidation states of Cr in [Cr(H$$_2$$O)$$_6$$]Cl$$_3$$, [Cr(C$$_6$$H$$_6$$)$$_2$$] and K$$_2$$[Cr(CN)$$_2$$(O)$$_2$$(O$$_2$$)(NH$$_3$$)], respectively, are:
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 54
The major product of the following reaction is: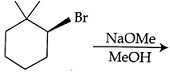
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 55
Phenol reacts with methyl chloroformate in the presence of NaOH to form product A. A reacts with Br$$_2$$ to form product B. A and B are respectively:
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 56
The major product formed in the following reaction is: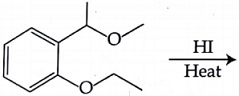
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 57
Phenol on treatment with $$CO_2$$ in the presence of NaOH followed by acidification produces compound X as the major product. X on treatment with $$(CH_3CO)_2O$$ in the presence of catalytic amount of $$H_2SO_4$$ produces:
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 58
The increasing order of basicity of the following compounds is: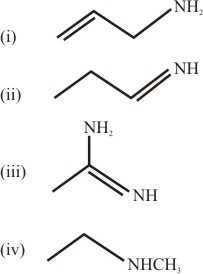
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 59
Glucose on prolonged heating with HI gives:
NTA JEE Main 8 April 2018 Offline - Chemistry - Question 60
The predominant form of histamine present in human blood is (pK$$_a$$, Histidine = 6.0):
.webp)
.webp)
.webp)
.webp)




