NTA JEE Main 8th April 2017 Online
For the following questions answer them individually
NTA JEE Main 8th April 2017 Online - Question 41
A metal M reacts with nitrogen gas to afford $$M_3N$$, $$M_3N$$ on heating at high temperature gives back M and on reaction with water produces a gas B. Gas B reacts with an aqueous solution of CuSO$$_4$$ to form a deep blue compound. M and B respectively are:
NTA JEE Main 8th April 2017 Online - Question 42
Consider the following ionization enthalpies of two elements 'A' and 'B'.
| Element | 1st (kJ/mol) | 2nd (kJ/mol) | 3rd (kJ/mol) |
|---|---|---|---|
| A | 899 | 1757 | 14847 |
| B | 737 | 1450 | 7731 |
Which of the following statements is correct?
NTA JEE Main 8th April 2017 Online - Question 43
Which of the following statements is not true about partition chromatography?
NTA JEE Main 8th April 2017 Online - Question 44
The IUPAC name of the following compound is: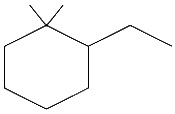
NTA JEE Main 8th April 2017 Online - Question 45
Which of the following compounds will not undergo Friedel Crafts reaction with benzene?
NTA JEE Main 8th April 2017 Online - Question 46
Identify the pollutant gases largely responsible for the discoloured and lustreless nature of marble of the Taj Mahal.
NTA JEE Main 8th April 2017 Online - Question 47
5 g of Na$$_2$$SO$$_4$$ was dissolved in $$x$$ g of H$$_2$$O. The change in freezing point was found to be 3.82°C. If Na$$_2$$SO$$_4$$ is 81.5% ionised, the value of $$x$$ is approximately: ($$K_f$$ for water = 1.86°C kg mol$$^{-1}$$) (molar mass of S = 32 g mol$$^{-1}$$ and that of Na = 23 g mol$$^{-1}$$)
NTA JEE Main 8th April 2017 Online - Question 48
Consider the following standard electrode potentials ($$E°$$ in volts) in aqueous solution:
| Element | $$M^{3+}/M$$ | $$M^+/M$$ |
|---|---|---|
| Al | $$-1.66$$ | $$+0.55$$ |
| Tl | $$+1.26$$ | $$-0.34$$ |
Based on these data, which of the following statements is correct?
NTA JEE Main 8th April 2017 Online - Question 49
What is the standard reduction potential ($$E°$$) for $$Fe^{3+} \to Fe$$?
Given that:
$$Fe^{2+} + 2e^- \to Fe$$; $$E°_{Fe^{2+}/Fe} = -0.47$$ V
$$Fe^{3+} + e^- \to Fe^{2+}$$; $$E°_{Fe^{3+}/Fe^{2+}} = +0.77$$ V
NTA JEE Main 8th April 2017 Online - Question 50
The rate of a reaction A doubles on increasing the temperature from 300 to 310 K. By how much, the temperature of reaction B should be increased from 300 K so that rate doubles if activation energy of the reaction B is twice to that of reaction A.


