NTA JEE Main 7th January 2020 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 26
The ammonia (NH$$_3$$) released on quantitative reaction of 0.6 g urea (NH$$_2$$CONH$$_2$$) with sodium hydroxide (NaOH) can be neutralized by
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 27
Within each pair of elements F and Cl, S and Se, and Li and Na, respectively, the elements that release more energy upon an electron gain are
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 28
The bond order and the magnetic characteristic of CN$$^-$$ are
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 29
Identify the correct labels of A, B and C in the following graph from the options given below: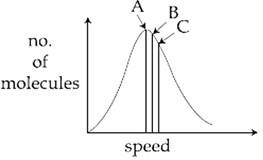
Root mean square speed (V$$_{rms}$$); most probable speed (V$$_{mp}$$); Average speed (V$$_{av}$$)
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 30
The redox reaction among the following is
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 31
Among statements (a) - (d), the correct ones are:
(a) Decomposition of hydrogen peroxide gives di-oxygen.
(b) Like hydrogen peroxide, compounds, such as KClO$$_3$$, Pb(NO$$_3$$) and NaNO$$_3$$ when heated liberate di-oxygen.
(c) 2-Ethylanthraquinone is useful for the industrial preparation of hydrogen peroxide.
(d) Hydrogen peroxide is used for the manufacture of sodium perborate.
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 32
The correct order of stability for the following alkoxides is:
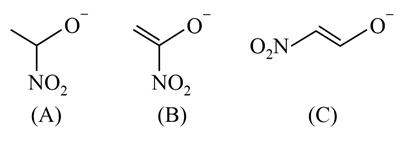
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 33
A chromatography column, packed with silica gel as stationary phase, was used to separate a mixture of compounds consisting of (A) benzanilide (B) aniline and (C) acetophenone. When the column is eluted with a mixture of solvents, hexane:ethyl acetate (20 : 80), the sequence of obtained compounds is:
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 34
Two open beakers one containing a solvent and the other containing a mixture of that solvent with a non volatile solute are together sealed in a container. Over time:
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 35
The equation that is incorrect is:
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 36
For the reaction $$2H_2(g) + 2NO(g) \rightarrow N_2(g) + 2H_2O(g)$$ the observed rate expression is, rate $$= k_f[NO]^2[H_2]$$. The rate expression for the reverse reaction is:
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 37
The refining method used when the metal and the impurities have low and high melting temperatures, respectively, is
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 38
In the following reactions, products (A) and (B), respectively, are:
NaOH + Cl$$_2$$ $$\rightarrow$$ (A) + side products (hot and conc.)
Ca(OH)$$_2$$ + Cl$$_2$$ $$\rightarrow$$ (B) + side products (dry)
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 39
The number of possible optical isomers for the complexes MA$$_2$$B$$_2$$ with sp$$^3$$ and dsp$$^2$$ hybridized metal atom, respectively, is:
Note: A and B are unidentate neutral and unidentate monoanionic ligands, respectively.
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 40
Among the statements (a) - (d), the incorrect ones are:
(a) Octahedral Co(III) complexes with strong field ligands have very high magnetic moments
(b) When $$\Delta_0 < P$$, the d-electron configuration of Co(III) in an octahedral complex is $$t_{eg}^4 e_g^2$$
(c) Wavelength of light absorbed by [Co(en)$$_3$$]$$^{3+}$$ is lower than that of [CoF$$_6$$]$$^{3-}$$
(d) If the $$\Delta_0$$ for an octahedral complex of Co(III) is 18,000 cm$$^{-1}$$, the $$\Delta_t$$ for its tetrahedral complex with the same ligand will be 16,000 cm$$^{-1}$$.
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 41
For the following reactions
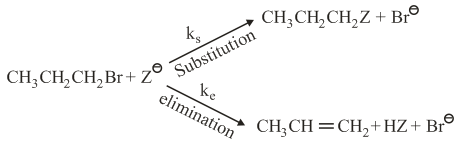
where:
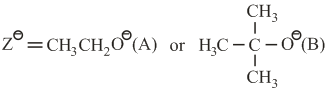
k$$_s$$ and k$$_e$$ are respectively the rate constants for substitution and elimination, and $$\mu = \frac{k_s}{k_e}$$, the correct option is
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 42
Consider the following reactions:
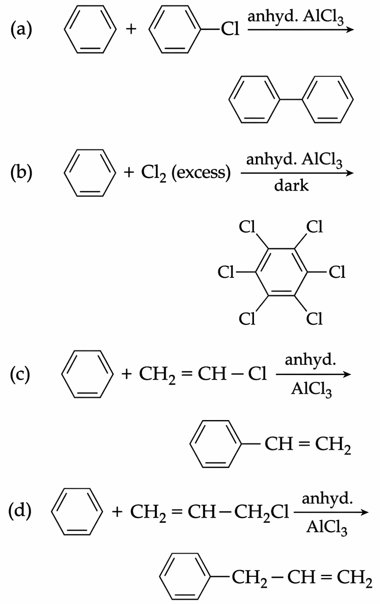
Which of these reactions are possible?
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 43
In the following reaction sequence, structures of A and B are, respectively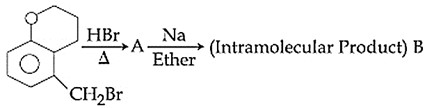
Cyclohexanone-CH$$_2$$Br $$\xrightarrow{\text{HBr}}$$ A $$\xrightarrow{\text{Na/Ether}}$$ (Intramolecular Product) B
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 44
In the following reaction sequence,
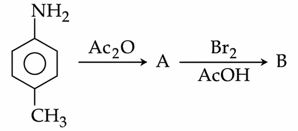
the major product B is
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 45
Which of the following statements is correct?
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 46
The standard heat of formation $$(\Delta_f H_{298}^0)$$ of ethane (in kJ/mol), if the heat of combustion of ethane, hydrogen and graphite are $$-1560$$, $$-393.5$$ and $$-286$$ kJ/mol, respectively is
789
456
123
0.-
Clear All
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 47
3 g of acetic acid is added to 250 mL of 0.1 M HCl and the solution made up to 500 mL. To 20 mL of this solution $$\frac{1}{2}$$ mL of 5M NaOH is added. The pH of the solution is
[Given: pKa of acetic acid = 4.75, molar mass of acetic acid 60 g/mol, log 3 = 0.4771, Neglect any changes in volume]
789
456
123
0.-
Clear All
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 48
Consider the following reactions:
NaCl + K$$_2$$Cr$$_2$$O$$_7$$ + H$$_2$$SO$$_4$$ (Conc.) $$\rightarrow$$ (A) + side products
(A) + NaOH $$\rightarrow$$ (B) + side products
(B) + H$$_2$$SO$$_4$$ + H$$_2$$O$$_2$$ (dilute) $$\rightarrow$$ (C) + side products
The sum of the total number of atoms in one molecule each of (A), (B) and (C) is
789
456
123
0.-
Clear All
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 49
The flocculation value of HCl for arsenic sulphide sol is 30 mmolL$$^{-1}$$. If H$$_2$$SO$$_4$$ is used for the flocculation of arsenic sulphide, the amount, in grams, of H$$_2$$SO$$_4$$ in 250 ml required for the above purpose is (molecular mass of H$$_2$$SO$$_4$$ = 98 g/mol)
789
456
123
0.-
Clear All
NTA JEE Main 7th January 2020 Shift 2 - Chemistry - Question 50
The number of sp$$^2$$ hybridised carbons present in "Aspartame" is
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




