NTA JEE Main 7th January 2020 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 26
Amongst the following statements, that which was not proposed by Dalton was:
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 27
The number of orbitals associated with quantum numbers $$n = 5$$, $$m_s = +\frac{1}{2}$$ is:
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 28
The electron gain enthalpy (in kJ/mol) of fluorine, chlorine, bromine and iodine, respectively, are
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 29
The dipole moments of CCl$$_4$$, CHCl$$_3$$ and CH$$_4$$ are in the order
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 30
The relative strength of the interionic/ intermolecular forces in a decreasing order is:
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 31
Oxidation number of potassium in K$$_2$$O, K$$_2$$O$$_2$$ and KO$$_2$$, respectively, is:
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 32
In comparison to the zeolite process for the removal of permanent hardness, the synthetic resin method is
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 33
A solution of m-chloroaniline, m-chlorophenol and m-chlorobenzoic acid in ethyl acetate was extracted initially with a saturated solution of NaHCO$$_3$$ to give fraction A. The left over organic phase was extracted with dilute NaOH solution to give fraction B. The final organic layer was labelled as fraction C. Fractions A, B and C, contain respectively:
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 34
The increasing order of pK$$_b$$ for the following compounds will be:
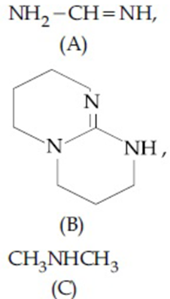
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 35
Consider the following reactions:
(a) $$(CH_3)_3CCH(OH)CH_3 \xrightarrow{\text{conc.} H_2SO_4}$$
(b) $$(CH_3)_2CHCH(Br)CH_3 \xrightarrow{\text{alc. KOH}}$$
(c) $$(CH_3)_2CHCH(Br)CH_3 \xrightarrow{(CH_3)_3O^\ominus K^\oplus}$$
(d)
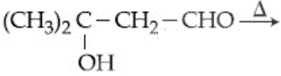
Which of the reaction(s) will not produce Saytzeff product?
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 36
At 35 $$^\circ$$C, the vapour pressure of CS$$_2$$ is 512 mmHg and that of acetone is 144 mmHg. A solution of CS$$_2$$ in acetone has a total vapour pressure of 600 mmHg. The false statement amongst the following is:
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 37
Given that the standard potentials (E$$^\circ$$) of Cu$$^{2+}$$/Cu and Cu$$^+$$/Cu are 0.34V and 0.522V respectively, the E$$^\circ$$ of Cu$$^{2+}$$/Cu$$^+$$ is:
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 38
The purest form of commercial iron is:
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 39
The atomic radius of Ag is closest to
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 40
The IUPAC name of the complex [Pt(NH$$_3$$)$$_2$$Cl(NH$$_2$$CH$$_3$$)]Cl is
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 41
The theory that can completely/properly explain the nature of bonding in [Ni(CO)$$_4$$] is:
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 42
1-methyl ethylene oxide when treated with an excess of HBr produces
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 43
What is the product of the following reaction?
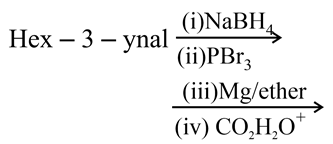
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 44
Consider the following reaction: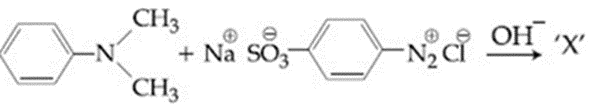
The product 'X' is used:
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 45
Match the following:
(i) Riboflavin (a) Beriberi
(ii) Thiamine (b) Scurvy
(iii) Pyridoxine (c) Cheilosis
(iv) Ascorbic acid (d) Convulsions
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 46
For the reaction:
A(l) $$\rightarrow$$ 2B(g)
$$\Delta U = 2.1$$ kcal, $$\Delta S = 20$$ cal K$$^{-1}$$ at 300K.
Hence $$\Delta G$$ in kcal is
789
456
123
0.-
Clear All
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 47
Two solutions, A and B, each of 100L was made by dissolving 4g of NaOH and 9.8g of H$$_2$$SO$$_4$$ in water, respectively. The pH of the resultant solution obtained from mixing 40L of solution A and 10L of solution B is (log 2 = 0.3)
789
456
123
0.-
Clear All
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 48
During the nuclear explosion, one of the products is $$^{90}$$Sr with half life of 6.93 years. If 1$$\mu$$g of $$^{90}$$Sr was absorbed in the bones of a newly born baby in place of Ca, how much time, in years, is required to reduce it by 90% if it is not lost metabolically
789
456
123
0.-
Clear All
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 49
Chlorine reacts with hot and concentrated NaOH and produces compounds (X) and (Y). Compound (X) gives white precipitate with silver nitrate solution. The average bond order between Cl and O atoms in (Y) is
789
456
123
0.-
Clear All
NTA JEE Main 7th January 2020 Shift 1 - Chemistry - Question 50
The number of chiral carbons in chloramphenicol is
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




