NTA JEE Main 6th September 2020 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 26
The average molar mass of chlorine is $$35.5\,\text{g mol}^{-1}$$. The ratio of $$^{35}\text{Cl}$$ to $$^{37}\text{Cl}$$ in naturally occurring chlorine is close to:
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 27
For a reaction $$4M(s) + nO_2(g) \to 2M_2O_n(s)$$. The free energy change is plotted as a function of temperature. The temperature below which the oxide is stable could be inferred from the plot as the point at which:
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 28
The value of $$K_c$$ is 64 at 800 K for the reaction
$$\text{N}_2(g) + 3\text{H}_2(g) \to 2\text{NH}_3(g)$$
The value of $$K_c$$ for the following reaction is:
$$\text{NH}_3(g) \to \frac{1}{2}\text{N}_2(g) + \frac{3}{2}\text{H}_2(g)$$
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 29
Dihydrogen of high purity (> 99.95%) is obtained through:
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 30
Match the following compounds (Column-I) with their uses (Column-II):
S. No. Column-I S.No. Column-II
(I) $$\text{Ca(OH)}_2$$ - (A) casts of statues
(II) NaCl - (B) white wash
(III) $$\text{CaSO}_4 \cdot \frac{1}{2}\text{H}_2\text{O}$$ - (C) antacid
(IV) $$\text{CaCO}_3$$ - (D) washing soda preparation
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 31
Match the following:
Test/Method Reagent
(i) Lucas Test (a) $$\text{C}_6\text{H}_5\text{SO}_2\text{Cl}$$ / aq. KOH
(ii) Dumas method (b) $$\text{HNO}_3$$ / $$\text{AgNO}_3$$
(iii) Kjeldahl's method (c) CuO / $$\text{CO}_3$$
(iv) Hinsberg Test (d) Conc. HCl and $$\text{ZnCl}_2$$
(e) $$\text{H}_2\text{SO}_4$$
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 32
The IUPAC name of the following compound is: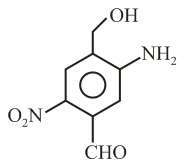
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 33
A crystal is made up of metal ions 'M1' and 'M2' and oxide ions. Oxide ions form a ccp lattice structure. The cation 'M1' occupies 50% of octahedral voids and the cation 'M2' occupies 12.5% of tetrahedral voids of oxide lattice. The oxidation numbers of 'M1' and 'M2' are respectively:
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 34
A set of solutions is prepared using 180 g of water as a solvent and 10 g of different non-volatile solutes A, B and C. The relative lowering of vapour pressure in the presence of these solutes are in the order [Given, molar mass of $$A = 100\,\text{g mol}^{-1}$$; $$B = 200\,\text{g mol}^{-1}$$; $$C = 10,000\,\text{g mol}^{-1}$$]
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 35
For the given cell; $$\text{Cu}(s)|\text{Cu}^{2+}(C_1\,M)||\text{Cu}^{2+}(C_2\,M)|\text{Cu}(s)$$
change in Gibbs energy $$(\Delta G)$$ is negative, it:
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 36
The element that can be refined by distillation is:
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 37
The reaction of NO with $$\text{N}_2\text{O}_4$$ at 250 K gives:
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 38
Reaction of an inorganic sulphite X with dilute $$\text{H}_2\text{SO}_4$$ generated compound Y. Reaction of Y with NaOH gives X. Further, the reaction of X with Y and water affords compound Z. Y and X, respectively, are:
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 39
Mischmetal is an alloy consisting mainly of:
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 40
For a $$d^4$$ metal ion in an octahedral field, the correct electronic configuration is:
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 41
The increasing order of the boiling points of the major products A, B and C of the following reactions will be: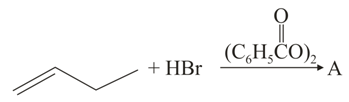
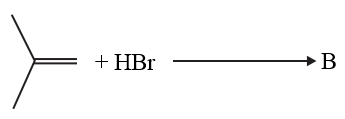
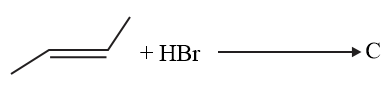
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 42
The correct match between Item-I (starting material) and Item-II (reagent) for the preparation of benzaldehyde is:
Item-I Item-II
(I) Benzene (P) HCl and $$\text{SnCl}_2$$, $$\text{H}_3\text{O}^+$$
(II) Benzonitrile (Q) $$\text{H}_2$$, $$\text{Pd-BaSO}_4$$, S and quinoline
(III) Benzoyl Chloride (R) Co, HCl and $$\text{AlCl}_3$$
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 43
Which of the following compounds can be prepared in good yield by Gabriel phthalimide synthesis?
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 44
The correct match between item-I and item-II is:
Item-I Item-II
(a) Natural rubber (I) 1,3-butadiene + styrene
(b) Neoprene (II) 1,3-butadiene
(c) Buna-N (III) chloroprene
(d) Buna-S (IV) Isoprene
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 45
Which one of the following statement is not true?
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 46
The atomic number of Unnilunium is______.
789
456
123
0.-
Clear All
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 47
If the solubility product of $$\text{AB}_2$$ is $$3.20 \times 10^{-11}\,\text{M}^3$$, then the solubility of $$\text{AB}_2$$ in pure water is ______ $$\times 10^{-4}\,\text{mol L}^{-1}$$ [Assuming that neither kind of ion reacts with water]
789
456
123
0.-
Clear All
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 48
The rate of a reaction decreased by 3.555 times when the temperature was changed from $$40\,^\circ\text{C}$$ to $$30\,^\circ\text{C}$$. The activation energy (in $$\text{kJ mol}^{-1}$$) of the reaction is______.
789
456
123
0.-
Clear All
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 49
For Freundlich adsorption isotherm, a plot of $$\log(x/m)$$ (y-axis) and $$\log p$$ (x-axis) gives a straight line, the intercept and slope for the line is 0.4771 and 2, respectively. The mass of gas, adsorbed per gram of adsorbent if the initial pressure is 0.04 atm is ______ $$\times 10^{-4}\,\text{g}$$.
789
456
123
0.-
Clear All
NTA JEE Main 6th September 2020 Shift 2 - Chemistry - Question 50
A solution of phenol in chloroform when treated with aqueous NaOH gives compound P as a major product. The mass percentage of carbon in P is______.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




