NTA JEE Main 6th September 2020 Shift 1
For the following questions answer them individually
NTA JEE Main 6th September 2020 Shift 1 - Question 41
The set that contains atomic numbers of only transition elements, is:
NTA JEE Main 6th September 2020 Shift 1 - Question 42
The lanthanoid that does NOT show +4 oxidation state is:
NTA JEE Main 6th September 2020 Shift 1 - Question 43
The species that has a spin-only magnetic moment of 5.9 BM, is: ($$T_d$$ = tetrahedral)
NTA JEE Main 6th September 2020 Shift 1 - Question 44
The increasing order of $$pK_b$$ values of the following compounds is: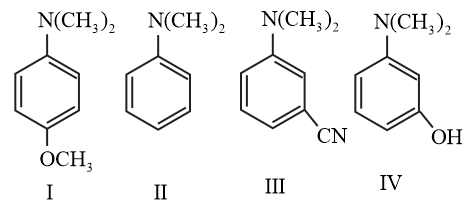
NTA JEE Main 6th September 2020 Shift 1 - Question 45
Consider the Assertion and Reason given below.
Assertion (A): Ethene polymerized in the presence of Ziegler Natta Catalyst at high temperature and pressure is used to make buckets and dustbins.
Reason (R): High density polymers are closely packed and are chemically inert. Choose the correct answer from the following:
NTA JEE Main 6th September 2020 Shift 1 - Question 46
A spherical balloon of radius $$3\,\text{cm}$$ containing helium gas has a pressure of $$48 \times 10^{-3}\,\text{bar}$$. At the same temperature, the pressure, of a spherical balloon of radius $$12\,\text{cm}$$ containing the same amount of gas will be............ $$\times 10^{-6}\,\text{bar}$$.
789
456
123
0.-
Clear All
NTA JEE Main 6th September 2020 Shift 1 - Question 47
In an estimation of bromine by Carius method, $$1.6\,\text{g}$$ of an organic compound gave $$1.88\,\text{g}$$ of AgBr. The mass percentage of bromine in the compound is........ (Atomic mass, Ag = 108, Br = 80 $$\text{g mol}^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 6th September 2020 Shift 1 - Question 48
The elevation of boiling point of $$0.10\,\text{m}$$ aqueous $$\text{CrCl}_3 \cdot x\text{NH}_3$$ solution is two times that of $$0.05\,\text{m}$$ aqueous $$\text{CaCl}_2$$ solution. The value of $$x$$ is............ [Assume 100% ionisation of the complex and $$\text{CaCl}_2$$, coordination number of Cr as 6, and that all $$\text{NH}_3$$ molecules are present inside the coordination sphere]
789
456
123
0.-
Clear All
NTA JEE Main 6th September 2020 Shift 1 - Question 49
Potassium chlorate is prepared by the electrolysis of KCl in basic solution
$$6\text{OH}^- + \text{Cl}^- \to \text{ClO}_3^- + 3\text{H}_2\text{O} + 6e^-$$. If only 60% of the current is utilized in the reaction, the time (rounded to the nearest hour) required to produce $$10\,\text{g}$$ of $$\text{KClO}_3$$ using a current of $$2\,\text{A}$$ is........ (Given: F = 96,500 C/mol; molar mass of $$\text{KClO}_3$$ = 122 g mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 6th September 2020 Shift 1 - Question 50
The number of $$\text{Cl} = \text{O}$$ bonds in perchloric acid is, $$n$$.........
789
456
123
0.-
Clear All


