NTA JEE Main 6th September 2020 Shift 1
For the following questions answer them individually
NTA JEE Main 6th September 2020 Shift 1 - Question 21
The density of a solid metal sphere is diameter. The maximum error in the density of the sphere is $$\left(\frac{x}{100}\right)\%$$. If the relative errors in measuring the mass and the diameter are $$6.0\%$$ and $$1.5\%$$ respectively, the value of $$x$$ is ___
789
456
123
0.-
Clear All
NTA JEE Main 6th September 2020 Shift 1 - Question 22
Two bodies of the same mass are moving with the same speed, but in different directions in a plane. They have a completely inelastic collision and move together thereafter with a final speed which is half of their initial velocities of the two bodies (in degree) is ___
789
456
123
0.-
Clear All
NTA JEE Main 6th September 2020 Shift 1 - Question 23
Initially a gas of diatomic molecules is contained in a cylinder of volume $$V_1$$ at a pressure $$P_1$$ and temperature $$250\,\text{K}$$. Assuming that $$25\%$$ of the molecules get dissociated causing a change in number of moles. The pressure of the resulting gas at temperature $$2000\,\text{K}$$, when contained in a volume $$2V_1$$ is given by $$P_2$$. The ratio $$P_2/P_1$$ is ___
789
456
123
0.-
Clear All
NTA JEE Main 6th September 2020 Shift 1 - Question 24
Suppose that intensity of a laser is $$\left(\frac{315}{\pi}\right)\,\text{W m}^{-2}$$. The rms electric field, in units of $$\text{V m}^{-1}$$ associated with this source is close to the nearest integer is ___ ($$\varepsilon_0 = 8.86 \times 10^{-12}\,\text{C}^2\,\text{N m}^{-2}$$; $$c = 3 \times 10^8\,\text{m s}^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 6th September 2020 Shift 1 - Question 25
A part of a complete circuit is shown in the figure. At some instant, the value of current I is $$1\,\text{A}$$ and it is decreasing at a rate of $$10^2\,\text{As}^{-1}$$. The value of the potential difference $$V_P - V_Q$$, (in volts) at that instant is___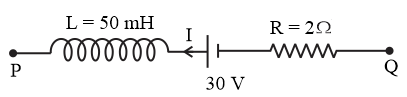
789
456
123
0.-
Clear All
NTA JEE Main 6th September 2020 Shift 1 - Question 26
A solution of two components containing $$n_1$$ moles of the 1st component and $$n_2$$ moles of the 2nd component is prepared. $$M_1$$ and $$M_2$$ are the molecular weights of component 1 and 2 respectively. If $$d$$ is the density of the solution in $$\text{g mL}^{-1}$$, $$C_2$$ is the molarity and $$x_2$$ is the mole fraction of the 2nd component, then $$C_2$$ can be expressed as:
NTA JEE Main 6th September 2020 Shift 1 - Question 27
The variation of equilibrium constant with temperature is given below:
Temperature: , Equilibrium Constant:
$$T_1 = 25^\circ\text{C}$$ $$K_1 = 10$$
$$T_2 = 100^\circ\text{C}$$, $$K_2 = 100$$
The values of $$\Delta H^\circ$$, $$\Delta G^\circ$$ at $$T_1$$ and $$\Delta G^\circ$$ at $$T_2$$ (in $$\text{kJ mol}^{-1}$$) respectively, are close to [use $$R = 8.314\,\text{JK}^{-1}\text{mol}^{-1}$$]
NTA JEE Main 6th September 2020 Shift 1 - Question 28
For the reaction
$$\text{Fe}_2\text{N}(s) + \frac{3}{2}\text{H}_2(g) \rightleftharpoons 2\text{Fe}(s) + \text{NH}_3(g)$$
NTA JEE Main 6th September 2020 Shift 1 - Question 29
Arrange the following solutions in the decreasing order of pOH:
(A) 0.01 M HCl
(B) 0.01 M NaOH
(C) 0.01 M $$\text{CH}_3\text{COONa}$$
(D) 0.01 M NaCl
NTA JEE Main 6th September 2020 Shift 1 - Question 30
Among the sulphates of alkaline earth metals, the solubilities of $$\text{BeSO}_4$$ and $$\text{MgSO}_4$$ in water, respectively, are:


