NTA JEE Main 6th April 2023 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 31
If the radius of the first orbit of hydrogen atom is $$a_0$$, then de Broglie's wavelength of electron in 3$$^{rd}$$ orbit is
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 32
Which one of the following elements will remain as liquid inside pure boiling water?
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 33
Group-13 elements react with O$$_2$$ in amorphous form to form oxides of type M$$_2$$O$$_3$$ (M = element). Which among the following is the most basic oxide?
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 34
During the reaction of permanganate with thiosulphate, the change in oxidation of manganese occurs by value of 3. Identify which of the below medium will favour the reaction.
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 35
The volume of 0.02 M aqueous HBr required to neutralize 10.0 mL of 0.01 M aqueous Ba(OH)$$_2$$ is (Assume complete neutralization)
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 36
Ion having highest hydration enthalpy among the given alkaline earth metal ions is:
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 37
Structures of BeCl$$_2$$ in solid state, vapour phase and at very high temperature respectively are:
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 38
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 39
The strongest acid from the following is
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 40
The group of chemicals used as pesticide is
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 41
The product, which is not obtained during the electrolysis of brine solution is
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 42
Consider the following reaction that goes from A to B in three steps as shown below:
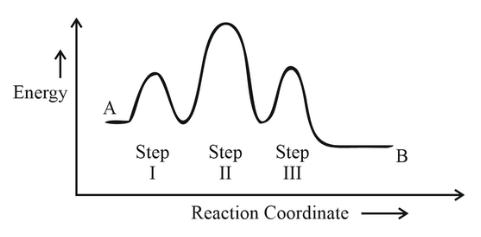
Choose the correct option

NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 43
The IUPAC name of K$$_3$$[Co(C$$_2$$O$$_4$$)$$_3$$] is:
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 44
Given below are two statements: one is labelled as 'Assertion A' and the other is labelled as 'Reason R'
Assertion A: In the complex Ni(CO)$$_4$$ and Fe(CO)$$_5$$, the metals have zero oxidation state.
Reason R: Low oxidation states are found when a complex has ligands capable of $$\pi$$-donor character in addition to the $$\sigma$$-bonding.
In the light of the above statements, choose the most appropriate answer from the options given below
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 45
Element not present in Nessler's reagent is
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 46
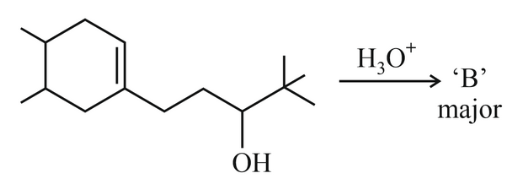
In the following reaction, 'B' is
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 47
Find out the major product from the following reaction.
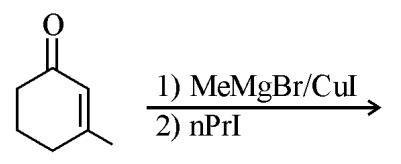
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 48
Given below are two statements:
Statement I: Morphine is a narcotic analgesic. It helps in relieving pain without producing sleep.
Statement II: Morphine and its derivatives are obtained from opium poppy.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 49
Formation of which complex, among the following, is not a confirmatory test of Pb$$^{2+}$$ ions
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 50
Match List-I with List-II.

Choose the correct answer from the options given below:
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 51
The number of species having a square planar shape from the following is
XeF$$_4$$, SF$$_4$$, SiF$$_4$$, BF$$_4^-$$, BrF$$_4^-$$, [Cu(NH$$_3$$)$$_4$$]$$^{2+}$$, [FeCl$$_4$$]$$^{2-}$$, [PtCl$$_4$$]$$^{2-}$$
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 52
In an ice crystal, each water molecule is hydrogen bonded to ______ neighbouring molecules.
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 53
Consider the following data
Heat of combustion of H$$_2$$(g) = -241.8 kJ mol$$^{-1}$$
Heat of combustion of C(s) = -393.5 kJ mol$$^{-1}$$
Heat of combustion of C$$_2$$H$$_5$$OH(l) = -1234.7 kJ mol$$^{-1}$$
The heat of formation of C$$_2$$H$$_5$$OH(l) is (-) ______ kJ mol$$^{-1}$$ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 54
The equilibrium composition for the reaction
$$PCl_3 + Cl_2 \rightleftharpoons PCl_5$$ at 298 K is given below:
[PCl$$_3$$]$$_{eq}$$ = 0.2 mol L$$^{-1}$$, [Cl$$_2$$]$$_{eq}$$ = 0.1 mol L$$^{-1}$$, [PCl$$_5$$]$$_{eq}$$ = 0.40 mol L$$^{-1}$$
If 0.2 mol of Cl$$_2$$ is added at the same temperature, the equilibrium concentrations of PCl$$_5$$ is ______ $$\times 10^{-2}$$ mol L$$^{-1}$$
Given: K$$_c$$ for the reaction at 298 K is 20
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 55
Number of crystal systems from the following where body centred unit cell can be found, is ______.
Cubic, tetragonal, orthorhombic, hexagonal, rhombohedral, monoclinic, triclinic
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 56
Consider the following pairs of solution which will be isotonic at the same temperature. The number of pairs of solutions is/are ______.
A. 1 M aq. NaCl and 2 M aq. urea
B. 1 M aq. CaCl$$_2$$ and 1.5 M aq. KCl
C. 1.5 M aq. AlCl$$_3$$ and 2 M aq. Na$$_2$$SO$$_4$$
D. 2.5 M aq. KCl and 1 M aq. Al$$_2$$(SO$$_4$$)$$_3$$
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 57
The standard reduction potentials at 295 K for the following half cells are given below:
NO$$_3^-$$ + 4H$$^+$$ + 3e$$^-$$ $$\rightarrow$$ NO(g) + 2H$$_2$$O E° = 0.97 V
V$$^{2+}$$(aq) + 2e$$^-$$ $$\rightarrow$$ V(s) E° = -1.19 V
Fe$$^{3+}$$(aq) + 3e$$^-$$ $$\rightarrow$$ Fe(s) E° = -0.04 V
Ag$$^+$$(aq) + e$$^-$$ $$\rightarrow$$ Ag(s) E° = 0.80 V
Au$$^{3+}$$(aq) + 3e$$^-$$ $$\rightarrow$$ Au(s) E° = 1.40 V
The number of metal(s) which will be oxidised by NO$$_3^-$$ in aqueous solution is ______.
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 58
The number of colloidal systems from the following, which will have 'liquid' as the dispersion medium, is ______.
Gem stones, paints, smoke, cheese, milk, hair cream, insecticide sprays, froth, soap lather
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 59
Among the following the number of compounds which will give positive iodoform reaction is ______.
(a) 1-Phenylbutan-2-one
(b) 2-Methylbutan-2-ol
(c) 3-Methylbutan-2-ol
(d) 1-Phenylethanol
(e) 3, 3-dimethylbutan-2-one
(f) 1-Phenylpropan-2-ol
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 2 - Chemistry - Question 60
Number of isomeric aromatic amines with molecular formula C$$_8$$H$$_{11}$$N, which can be synthesized by Gabriel Phthalimide synthesis is ______.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




