NTA JEE Main 6th April 2023 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 31
For a concentrated solution of a weak electrolyte ($$K_{eq}$$ = equilibrium constant) A$$_2$$B$$_3$$ of concentration 'C', the degree of dissociation '$$\alpha$$' is
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 32
Which of the following options are correct for the reaction?
$$2Au(CN)_2 ^-(aq) + Zn(s) \rightarrow 2Au(s) + Zn(CN)_4 ^{2-}(aq)$$
A. Redox reaction
B. Displacement reaction
C. Decomposition reaction
D. Combination reaction
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 33
Strong reducing and oxidizing agents among the following, respectively, are
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 34
Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Loss of electron from hydrogen atom results in nucleus of ~$$1.5 \times 10^{-3}$$ pm size.
Reason R: Proton H$$^+$$ always exists in combined form.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 35
The setting time of Cement is increased by adding
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 36
Match List-I with List-II.
List-I (Element detected) List-II (Reagent used/Product formed)
A. Nitrogen I. Na$$_2$$FeCN$$_5$$NO
B. Sulphur II. AgNO$$_3$$
C. Phosphorus III. Fe$$_4$$FeCN$$_{6_{3}}$$
D. Halogen IV. NH$$_{4_{2}}$$MoO$$_4$$
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 37
The possibility of photochemical smog formation is more at
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 38
A compound is formed by two elements X and Y. The element Y forms cubic close packed arrangement and those of element X occupy one third of the tetrahedral voids. What is the formula of the compound?
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 39
The standard electrode potential of M$$^+$$/M in aqueous solution does not depend on
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 40
Match List I with List II
LIST I - Enzymatic reaction LIST II - Enzyme
A. Sucrose $$\rightarrow$$ Glucose and Fructose I. Zymase
B. Glucose $$\rightarrow$$ ethyl alcohol and CO$$_2$$ II. Pepsin
C. Starch $$\rightarrow$$ Maltose III. Invertase
D. Proteins $$\rightarrow$$ Amino acids IV. Diastase
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 41
The difference between electron gain enthalpies will be maximum between:
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 42
Match List I with List II
List I (Oxide) List II (Type of bond)
A. N$$_2$$O$$_4$$ I. 1 N=O bond
B. NO$$_2$$ II. 1 N-O-N bond
C. N$$_2$$O$$_5$$ III. 1 N-N bond
D. N$$_2$$O IV. 1 N=N/N$$\equiv$$N bond
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 43
Match List-I with List-II.
List-I (Name of reaction) List-II (Reagent used)
A. Hell-Volhard Zelinsky reaction I. NaOH + I$$_2$$
B. Iodoform reaction II. (i) CrO$$_2$$Cl$$_2$$, CS$$_2$$ (ii) H$$_2$$O
C. Etard reaction III. (i) Br$$_2$$/red phosphorus (ii) H$$_2$$O
D. Gatterman-Koch reaction IV. CO, HCl, anhyd. AlCl$$_3$$
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 44
Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: The spin only magnetic moment value for Fe(CN)$$_6^{3-}$$ is 1.74 BM, whereas for [Fe(H$$_2$$O)$$_6$$]$$^{3+}$$ is 5.92 BM.
Reason B: In both complexes, Fe is present in +3 oxidation state.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 45
For the reaction

The correct statement is
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 46
The major products A and B from the following reactions are:
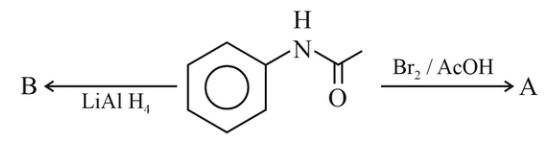
(1)
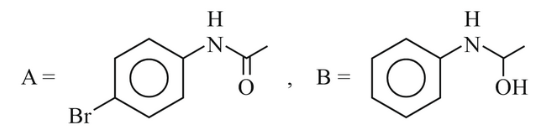
(2)
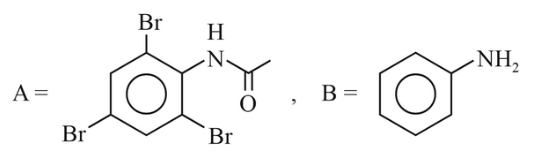
(3)
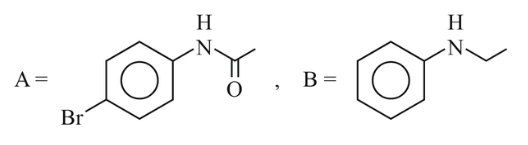
(4)
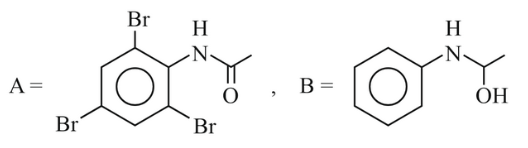
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 47
The major product formed in the following reaction is
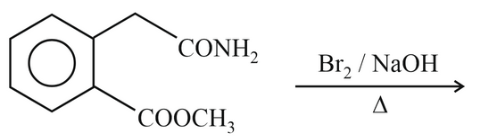
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 48
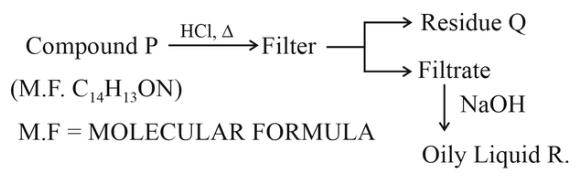
Compound P is neutral, Q gives effervescence with NaHCO$$_3$$ while R reacts with Hinsberg's reagent to give solid soluble in NaOH. Compound P is
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 49
Polymer used in orlon is:
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 50
Match List I and List II
List I (Vitamin) List II (Deficiency disease)
A. Vitamin A I. Beri-Beri
B. Thiamine II. Cheilosis
C. Ascorbic acid III. Xerophthalmia
D. Riboflavin IV. Scurvy
Choose the correct answer from the options given below
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 51
If 5 moles of BaCl$$_2$$ is mixed with 2 moles of Na$$_3$$PO$$_4$$, the maximum number of moles of Ba$$_3$$(PO$$_4$$)$$_2$$ formed is ______ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 52
The wavelength of an electron of kinetic energy $$4.50 \times 10^{-29}$$ J is ______ $$\times 10{-5}$$ m. (Nearest integer)
Given: mass of electron is $$9 \times 10{-31}$$ kg, h = $$6.6 \times 10{-34}$$ Js
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 53
The number of species from the following which have square pyramidal structure is ______
PF$$_5$$, BrF$$_4^-$$, IF$$_5$$, BrF$$_5$$, XeOF$$_4$$, ICl$$_4^-$$
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 54
The value of log K for the reaction A $$\rightleftharpoons$$ B at 298 K is ______. (Nearest integer)
Given: $$\Delta H° = -54.07$$ kJ mol$$^{-1}$$, $$\Delta S° = 10$$ J K$$^{-1}$$ mol$$^{-1}$$
(Taken $$2.303 \times 8.314 \times 298 = 5705$$)
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 55
Consider the graph of Gibbs free energy G vs extent of reaction. The number of statement/s from the following which are true with respect to points (a), (b) and (c) is ______
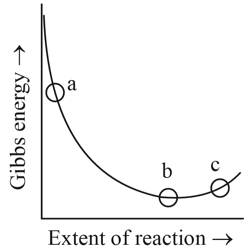
A. Reaction is spontaneous at (a) and (b)
B. Reaction is at equilibrium at point (b) and non-spontaneous at point (c)
C. Reaction is spontaneous at (a) and non-spontaneous at (c)
D. Reaction is non-spontaneous at (a) and (b)
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 56
Number of bromo derivatives obtained on treating ethane with excess of Br$$_2$$ in diffused sunlight is ______
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 57
Mass of Urea NH$$_2$$CONH$$_2$$ required to be dissolved in 1000 g of water in order to reduce the vapour pressure of water by 25% is ______ g. (Nearest integer)
Given: Molar mass of N, C, O and H are 14, 12, 16 and 1 g mol$$^{-1}$$ respectively.
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 58
For the adsorption of hydrogen on platinum, the activation energy is 30 kJ mol$$^{-1}$$ and for the adsorption of hydrogen on nickel, the activation energy is 41.4 kJ mol$$^{-1}$$. The logarithm of the ratio of the rates of chemisorption on equal areas of the metals at 300 K is ______ (Nearest integer)
Given: ln10 = 2.3, R = 8.3 J K$$^{-1}$$ mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 59
In ammonium-phosphomolybdate, the oxidation state of Mo is +______
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 1 - Chemistry - Question 60
Number of ambidentate ligands in a representative metal complex M(en)(SCN)$$_4$$ is ______. [en = ethylenediamine]
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




