NTA JEE Main 6th April 2014 Offline
For the following questions answer them individually
NTA JEE Main 6th April 2014 Offline - Question 31
The ratio of masses of oxygen and nitrogen in a particular gaseous mixture is 1 : 4. The ratio of number of their molecules is:
NTA JEE Main 6th April 2014 Offline - Question 32
The correct set of four quantum numbers for the valence electrons of rubidium atom ($$Z = 37$$) is:
NTA JEE Main 6th April 2014 Offline - Question 33
Which one of the following properties is not shown by NO?
NTA JEE Main 6th April 2014 Offline - Question 34
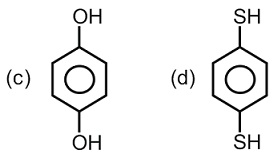
For which of the following molecule significant $$\mu \neq 0$$?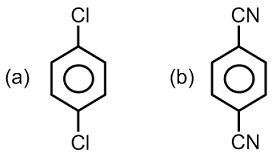
NTA JEE Main 6th April 2014 Offline - Question 35
If Z is the compressibility factor, then Van der Waal's equation at low pressure can be written as:
NTA JEE Main 6th April 2014 Offline - Question 36
For complete combustion of ethanol, $$C_2H_5OH(l) + 3O_2(g) \to 2CO_2(g) + 3H_2O(l)$$, the amount of heat produced as measured in bomb calorimeter, is 1364.47 kJ mol$$^{-1}$$ at 25°C. Assuming ideality the Enthalpy of combustion, $$\Delta_cH$$, for the reaction will be: ($$R = 8.314$$ kJ mol$$^{-1}$$)
NTA JEE Main 6th April 2014 Offline - Question 37
For the reaction $$SO_2(g) + \frac{1}{2}O_2(g) \rightleftharpoons SO_3(g)$$, if $$K_P = K_C(RT)^x$$ where the symbols have usual meaning, then the value of x is: (assuming ideality)
NTA JEE Main 6th April 2014 Offline - Question 38
In which of the following reactions $$H_2O_2$$ acts as a reducing agent?
(a) $$H_2O_2 + 2H^+ + 2e^- \to 2H_2O$$
(b) $$H_2O_2 - 2e^- \to O_2 + 2H^+$$
(c) $$H_2O_2 + 2e^- \to 2OH^-$$
(d) $$H_2O_2 + 2OH^- - 2e^- \to O_2 + 2H_2O$$
NTA JEE Main 6th April 2014 Offline - Question 39
For the estimation of nitrogen, 1.4 g of an organic compound was digested by the Kjeldahl method and the evolved ammonia was absorbed in 60 ml of $$\frac{M}{10}$$ sulphuric acid. The unreacted acid required 20 ml of $$\frac{M}{10}$$ sodium hydroxide for complete neutralization. The percentage of nitrogen in the compound is:
NTA JEE Main 6th April 2014 Offline - Question 40
Considering the basic strength of amines in an aqueous solution, which one has the smallest pK$$_b$$ value?


