NTA JEE Main 5th September 2020 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 26
The correct statement about probability density (except at infinite distance from nucleus) is:
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 27
The correct order of the ionic radii of $$\text{O}^{2-}$$, $$\text{N}^{3-}$$, $$\text{F}^-$$, $$\text{Mg}^{2+}$$, $$\text{Na}^+$$ and $$\text{Al}^{3+}$$ is:
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 28
The increasing order of boiling points of the following compounds is: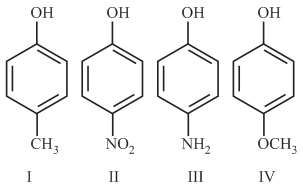
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 29
The compound that has the largest $$H-M-H$$ bond angle ($$M = N, O, S, C$$) is:
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 30
Lattice enthalpy and enthalpy of solution of NaCl are $$788\,\text{kJ mol}^{-1}$$ and $$4\,\text{kJ mol}^{-1}$$, respectively. The hydration enthalpy of $$NaCl$$ is:
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 31
Hydrogen peroxide, in the pure state, is:
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 32
The one that is NOT suitable for the removal of permanent hardness of water is:
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 33
Among the following compounds, geometrical isomerism is exhibited by:
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 34
The final major product of the following reaction is:
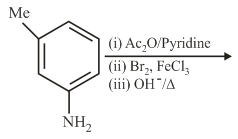
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 35
The major product formed in the following reaction is:
$$\text{CH}_2\text{CH} = \text{CHCH(CH}_3)_2 \xrightarrow{\text{HBr}}$$
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 36
An element crystallises in a face-centred cubic (fcc) unit cell with cell edge $$a$$. The distance between the centres of two nearest octahedral voids in the crystal lattice is:
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 37
The variation of molar conductivity with concentration of an electrolyte (X) in aqueous solution is shown in the given figure.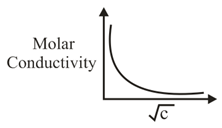
The electrolyte X is:
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 38
The rate constant $$(k)$$ of a reaction is measured at different temperature $$(T)$$, and the data are plotted in the given figure. The activation energy of the reaction in $$\text{kJ mol}^{-1}$$ is: (R is gas constant)
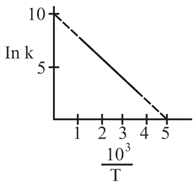
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 39
Adsorption of a gas follows Freundlich adsorption isotherm. If $$x$$ is the mass of the gas adsorbed on mass $$m$$ of the adsorbent, the correct plot of $$\frac{x}{m}$$ versus $$p$$ is:
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 40
Boron and silicon of very high purity can be obtained through:
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 41
Reaction of ammonia with excess $$Cl_2$$ gives:
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 42
Consider the complex ions, trans-$$[\text{Co(en)}_2\text{Cl}_2]^+$$ (A) and cis-$$[\text{Co(en)}_2\text{Cl}_2]^+$$ (B). The correct statement regarding them is:
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 43
The major product of the following reaction is:
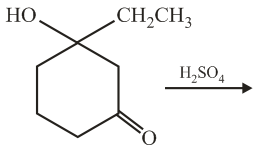
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 44
Which one of the following polymers is not obtained by condensation polymerisation?
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 45
The following molecule acts as an:
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 46
For a dimerization reaction,
$$2A(g) \to A_2(g)$$
at 298K, $$\Delta U^- = -20\,\text{kJ mol}^{-1}$$, $$\Delta S^- = -30\,\text{JK}^{-1}\text{mol}^{-1}$$, then the $$\Delta G^-$$ will be .....J.
789
456
123
0.-
Clear All
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 47
For a reaction $$X + Y = 2Z$$, $$1.0\,\text{mol}$$ of X, $$1.5\,\text{mol}$$ of Y and $$0.5\,\text{mol}$$ of Z were taken in a 1L vessel and allowed to react. At equilibrium, the concentration of Z was $$1.0\,\text{mol L}^{-1}$$. The equilibrium constant of the reaction is $$\frac{x}{15}$$. The value of x is.......
789
456
123
0.-
Clear All
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 48
The volume, in mL, of $$0.02\,\text{M}\,\text{K}_2\text{Cr}_2\text{O}_7$$ solution required to react with $$0.288\,\text{g}$$ of ferrous oxalate in acidic medium is............ (Molar mass of Fe = $$56\,\text{g mol}^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 49
Considering that $$\Delta_0 > P$$, the magnetic moment (in BM) of $$[\text{Ru}(\text{H}_2\text{O})_6]^{2+}$$ would be
789
456
123
0.-
Clear All
NTA JEE Main 5th September 2020 Shift 2 - Chemistry - Question 50
The number of chiral carbons present in sucrose is..........
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




